
(a)
Interpretation:
The balanced chemical equations for the given three steps processes involved in the production of high purity silicon needs to be determined.
Concept introduction:
The consecutive reactions take place one after another in a sequence to produce the final product. The simultaneous reactions involve the independent reaction of two or more substances reacting separately in another reaction at the same time.
The number of moles of a substance is related to mass and molar mass as follows:
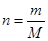
Here, m is mass and M is molar mass.
(b)
Interpretation:
The masses of carbon, chlorine and hydrogen required for production of per kilogram of high-purity silicon needs to be determined.
Concept introduction:
The consecutive reactions take place one after another in a sequence to produce the final product. The simultaneous reactions involve the independent reaction of two or more substances reacting separately in another reaction at the same time.
The number of moles of a substance is related to mass and molar mass as follows:
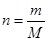
Here, m is mass and M is molar mass.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Mastering Chemistry With Pearson Etext -- Standalone Access Card -- For General Chemistry: Principles And Modern Applications (11th Edition)
- Complete and balance the following chemical equations: (a) hardening of plaster containing slaked lime. Ca(OH)2+CO2 (b) removal of sulfur dioxide from the ?ue gas of power plants. CaO+SO2 (c) the reaction of baking powder that produces carbon dioxide gas and causes bread to rise. NaHCO3+NaH2PO4arrow_forwardPhosphorus occurs naturally in the form of fluorapatite, CaF2 3Ca3(PO4)2. The dot indicates 1 part CaF2 to 3 parts Ca3(PO4)2. This mineral is reacted with an aqueous solution of H2SO4 in the preparation of a fertilizer. The products are phosphoric acid, hydrogen fluoride, and gypsum, CaSO4 2H2O. Write the balanced equation describing this process.arrow_forwardConsider the iron alloy described in Question 19. Suppose it is desired to prepare 1.00 kg of this alloy, what mass of each component would be necessary?arrow_forward
- Complete and balance the equations of the following reactions, each of which could be used to remove hydrogen sulfide from natural gas: (a) Ca(OH)2(s)+H2S(g) (b) Na2CO3(aq)+H2S(g)arrow_forwardDescribe how ultrapure silicon can be produced from sand.arrow_forwardThe extraction of aluminum metal from the aluminum hydroxide found in bauxite by the Hall-Héroult process is one of the most remarkable success stories of 19th century chemistry, turning aluminum from a rare and precious metal into the cheap commodity it is today. In the first step, aluminum hydroxide reacts to form alumina Al2O3 and water: 2AlOH3s + Al2O3s -> 3H2Og In the second step, alumina Al2O3 and carbon react to form aluminum and carbon dioxide: 2Al2O3s + 3Cs + 4Als -> 3CO2g Suppose the yield of the first step is 83.% and the yield of the second step is 78.% . Calculate the mass of aluminum hydroxide required to make 7.0kg of aluminum. Be sure your answer has a unit symbol, if needed, and is rounded to the correct number of significant digits.arrow_forward
- Halon 1301 (CF3Br) is used for Class B and C fires. It inhibits fire by reacting with the following EXCEPT with ____________. carbon dioxide B. halogen C. hydrocarbons D. oxygen Corrosion of iron is a combination reaction between Fes atoms and O2g molecules to form rust. Which of the following is the result of this reaction? FeO B. Fe2 C. Fe3O2 D. FeO3arrow_forward"Native," or elemental copper can be found in nature, but most copper is mined as oxide or sulfide minerals. Chalcopyrite (CuFeS2) is one copper mineral that can be converted to elemental copper in a series of chemical steps. Reacting chalcopyrite with oxygen at high temperature produces a mixture of copper sulfide and iron oxide. The iron oxide is separated from CuS by reaction with sand (SiO2). CuS is converted to Cu2S in the process and the Cu2S is burned in air to produce Cu and SO2:2CuFeS2+3O22CuS+2FeO+2SO2 2FeO+2SiO22FeSiO32CuSCu2S+SCu2S+S+2O22Cu+2SO2 An average copper penny minted in the 1960s contained about 3.000 g of copper. How much chalcopyrite had be mined to produce 100 pennies? How much chalcopyrite had to be mined to produce 100 pennies if reaction 1 had a percent yield of 83.00 % and all other reaction steps had yield of 100%? How much chalcopyrite had be mined to produce 100 pennies if each reactions 1, 3, and 4 proceeded in 83.00 % yield?arrow_forward"Native," or elemental copper can be found in nature, but most copper is mined as oxide or sulfide minerals. Chalcopyrite (CuFeS2) is one copper mineral that can be converted to elemental copper in a series of chemical steps. Reacting chalcopyrite with oxygen at high temperature produces a mixture of copper sulfide and iron oxide. The iron oxide is separated from CuS by reaction with sand (SiO2). CuS is converted to Cu2S in the process and the Cu2S is burned in air to produce Cu and SO2: 2CuFeS2+3O2=2CuS+2FeO+2SO2 2FeO+2SiO2=2FeSiO3 2CuS=Cu2S+S Cu2S+S+2O2=2Cu+2SO2 a) An average copper penny minted in the 1960s contained about 3.000 g of copper. How much chalcopyrite had be mined to produce 100 pennies? b) How much chalcopyrite had to be mined to produce 100 pennies if reaction 1 had a percent yield of 78.00 % and all other reaction steps had yield of 100%? c) How much chalcopyrite had be mined to produce 100 pennies if each reaction involving copper proceeded in 78.00 % yield?arrow_forward
- The Le Blanc Process for the manufacture of soda ash encompasses the four steps: NaCl + H2SO4 = NaHSO4 + HClNaHSO4 + NaCl = Na2SO4 + HClNa2SO4 + C = Na2S + CO2Na2S + CaCO3 = Na2CO3 + CaS How many tonnes of soda ash are produced per 5 tonnes of salt? How many kg of C are required?arrow_forwardElectric generating plants transport large amounts of hot water through metal pipes, and oxygen dissolved in the water can cause a major corrosion problem. Hydrazine (N2H4) added to the water avoids the problem by reacting with the oxygen: N2H4(aq) + O2(g) --> N2(g) + 2 H2O(l) About 4 × 107 kg of hydrazine is produced every year by reacting ammonia with sodium hypochlorite in the Raschig process: 2 NH3(aq) + NaOCl(aq) --> N2H4(aq) + NaCl(aq) + H2O(l) ΔrH° = −151 kJ (a) If ΔfH° of NaOCl(aq) = −346 kJ/mol, find ΔfH° of N2H4(aq) given that ΔfH° [NH3(aq)] = −80.83 kJ/mol, ΔfH° [NaOCl(aq)] = −346 kJ/mol, ΔfH° [Na+(aq)] = −239.66 kJ/mol, ΔfH° [Cl−(aq)] = −167.46 kJ/mol and ΔfH° [H2O(l)]= −285.840 kJ/mol. What is the heat released when aqueous N2H4 is added to 7.00 × 103 L of plant water that is 2.50 ×10−4 mol/L O2? Enter your answer in scientific notation. (Thanks in advance :))arrow_forwardMany important metals occur as sulfide, arsenide, and antimonide minerals, especially in the Sudburymineral complex. The first step in processing these ores involves “roasting” the ore in air to produce the metalor metal oxide, along with nonmetal oxides that can be serious pollutants if not trapped.Suppose that you roast 1.00 kg of the mineral maucherite, Ni11As8. The balanced equation for thereaction is:2 Ni11As8 + 31 O2 → 22 NiO + 8 As2O5(a) How many kg of NiO can be produced?(b) How many kg of the pollutant As2O5 can be produced?(c) How many liters of air at 25°C are required for roasting? Assume that air contains 23% O2 by mass,and that the density of air at 25°C is 1.2 g/L.Molar masses: Ni11As8 1245.0 g/mol NiO 74.69 g/mol As2O5 229.84 g/molarrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





