
(a)
Prove that the reflection coefficient for the given condition is
(a)
Answer to Problem 59CP
Proof for the reflection coefficient for the given condition is
Explanation of Solution
Write the Schrodinger’s equation.
Here,
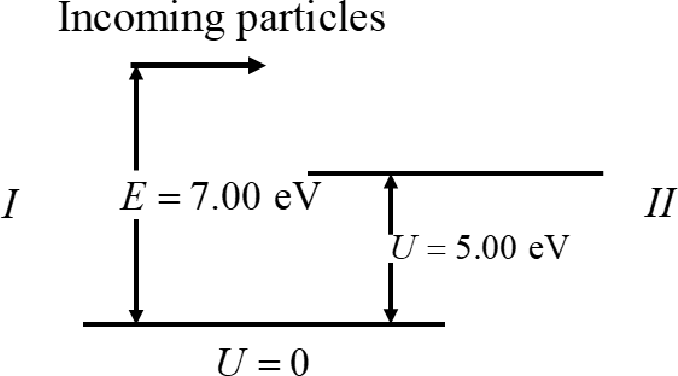
The solutions of the equation I for the region I in the above figure is
The solutions of the equation I for the region II in the above figure is
Here,
Check the solution for the region I satisfies the equation I.
The above equation will be true only if the
Here,
Rewrite the above equation to find the value of
Therefore the equation I is satisfied for the region I.
Check the equation I for the region II.
The above equation will be true only if the
Rewrite the above equation to find the value of
Therefore the equation I is satisfied for the region II.
Then apply the boundary conditions such as matching the function and derivatives at x=0.
From the above equations,
Write the equation for probability.
Conclusion:
Substitute equation VI in VII.
Therefore, the proof for the reflection coefficient for the given condition is
(b)
Find the probability of particle being reflected.
(b)
Answer to Problem 59CP
The probability of particle being reflected is
Explanation of Solution
Write the equation for ration of
Conclusion:
Substitute
Substitute
Therefore, the probability of particle being reflected is
(c)
Find the probability of particle being transmitted.
(c)
Answer to Problem 59CP
The probability of particle being transmitted is
Explanation of Solution
Write the equation for probability of transmitted.
Conclusion:
Substitute
Therefore, the probability of particle being transmitted is
Want to see more full solutions like this?
Chapter 41 Solutions
Physics For Scientists And Engineers With Modern Physics, 9th Edition, The Ohio State University
- An electron in a box is in the ground state with energy 2.0 eV. (a) Find the width of the box. (b) How much energy is needed to excite the electron to its first excited state? (c) If the electron makes a transition from an excited state to the ground state with the simultaneous emission of 30.0-eV photon, find the quantum number of the excited state?arrow_forwardA quantum mechanical oscillator vibrates at a frequency of 250.0 THz. What is the minimum energy of radiation it can emit?arrow_forwardA 600-nm light falls on a photoelectric surface and electrons with the maximum kinetic energy of 0.17 eV are emitted. Determine (a) the work function and (b) the cutoff frequency of the surface. (c) What is the stopping potential when the surface is illuminated with light of wavelength 400 nm?arrow_forward
- What speed does an F2 molecule have if it has the same momentum as a photon of wavelength 225 nm?arrow_forwardA 4.00-g particle confined to a box of length L has a speed of 1.00 mm/s. (a) What is the classical kinetic energy of the particle? (b) If the energy of the first excited state (n = 2) is equal to the kinetic energy found in part (a), what is the value of L? (c) Is the result found in part (b) realistic? Explain.arrow_forwardA paramecium is covered with motile hairs called cilia that propel it at a speed of 1 mm/s. If the paramecium has a volume of 2 x 10-13 m3 and a density equal to that of water, what is its de Broglie wavelength when in motion? What fraction of the paramecium’s 150 μm length does this wavelength represent?arrow_forward
 Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning




