
(a) In an HCl molecule, take the Cl atom to be the isotope 35Cl. The equilibrium separation of the H and Cl atoms is 0.127 46 nm. The
(a)
The longest wavelength in the rotational spectrum of HCl molecule when
Answer to Problem 11P
The longest wavelength in the rotational spectrum of
Explanation of Solution
Write the equation for the smallest energy difference between the rotational energy levels.
Here,
Write the equation for the wavelength.
Here,
Substitute equation (I) in the equation (II) and substitute
Write the equation for the reduced mass.
Here,
Write the equation for moment of inertia.
Here,
Conclusion:
Substitute equation (V) in equation (III).
Substitute
Substitute
Here,
Substitute
Thus, the longest wavelength in the rotational spectrum of HCl molecule when
(b)
The longest wavelength in the rotational spectrum of HCl molecule when
Answer to Problem 11P
The longest wavelength in the rotational spectrum of HCl molecule when
Explanation of Solution
From the equation (IV) for the reduced mass.
From the equation (VI) for wavelength.
Conclusion:
Substitute
Here,
Substitute
Thus, the longest wavelength in the rotational spectrum of HCl molecule when
(c)
The separation in wavelength between the doublet lines for the longest wavelength.
Answer to Problem 11P
The separation in wavelength between the doublet lines for the longest wavelength is
Explanation of Solution
Write the equation for the separation in wavelength between the doublet lines for the longest wavelength.
Here,
Conclusion:
Substitute
Thus, the separation in wavelength between the doublet lines for the longest wavelength is
Want to see more full solutions like this?
Chapter 42 Solutions
Physics for Scientists and Engineers with Modern Physics
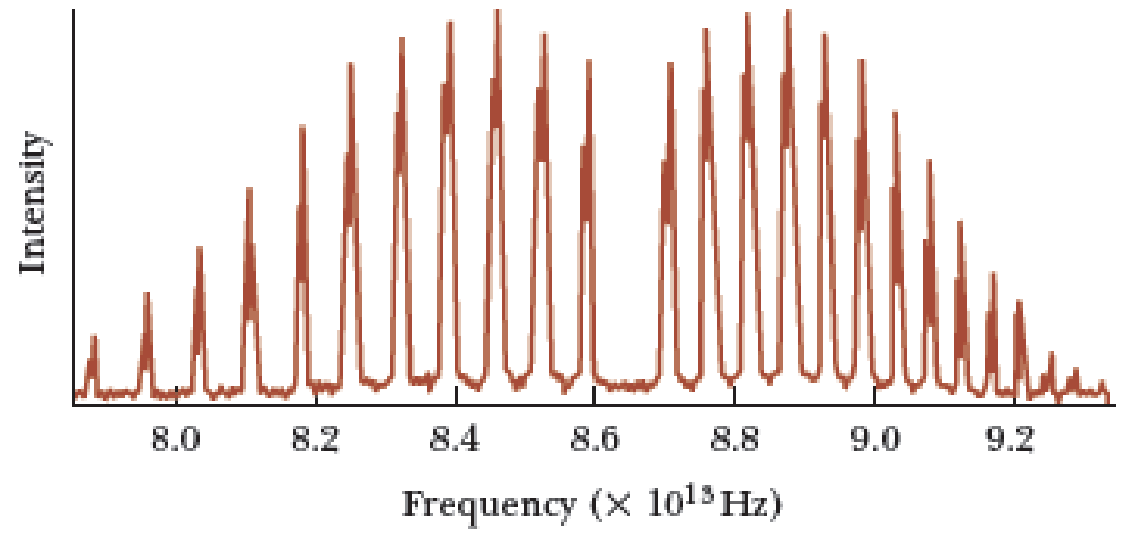
- Consider the HCl molecule, which consists of a hydrogen atom of mass 1 u bound to a chlorine atom of mass 35 u. The equilibrium separation between the atoms is 0.128 nm, and it requires 0.15 eV of work to increase or decrease this separation by 0.01 nm. (a) Calculate the four lowest rotational energies (in eV) that are possible, assuming the molecule rotates rigidly. (b) Find the molecules spring constant and its classical frequency of vibration. (Hint: Recall that U=12Kx2.) (c) Find the two lowest vibrational energies and the classical amplitude of oscillation corresponding to each of these energies. (d) Determine the longest wavelength radiation that the molecule can emit in a pure rotational transition and in a pure vibrational transition.arrow_forwardThe Madelung constant for the NaCl structure may be found by summing an infinite alternating series of terms giving the electrostatic potential energy between an Na+ ion and its 6 nearest Cl neighbors, its 12 next-nearest N+ neighbors, and so on (see Fig. 12.1a). (a) From this expression, show that the first three terms of the infinite series for the Madelung constant for the NaCl structure yield = 2.13. (b) Does this infinite series converge rapidly? Calculate the fourth term as a check.arrow_forwardThe equilibrium separation between the two ions in the KCl molecule is 0.267 nm. (a) Assuming that the K+ and Cl- ions are point particles, compute the electric dipole moment of the molecule. (b) Compute the ratio of your result in (a) to the measured electric dipole moment of 5.41 x 10-29 C*m. This ratio is known as the fractional ionic character of the molecular bond.arrow_forward
- Consider a CO molecule that is initially in the ground state of n = 0, l = 0. If the energy of a vibrational transition from the n = 0 state to the n = 1 state in CO could instead be absorbed in a rotational transition, what would be the value of l for the final state?arrow_forwardThe figure above shows the absorption spectrum of the molecule HBr. Following the basic procedures of Section 9.6, find:(a) the energy of the “missing” transition;(b) the effective force constant k;(c) the rotational spacing 2B. Estimate the value of the rotational spacing expected for HBr and compare with the value deduced from the spectrum. Why are there only single lines and not double lines as in the case of HCl?arrow_forwardShow that the moment of inertia of a diatomic molecule composed of atoms of masses mA and mB and bond length R is equal to meffR2, where meff = mAmB/(mA+mB).arrow_forward
- Show that the moment of inertia of a diatomic molecule composed of atoms of masses mA and mB and bond length R is equal to meffR2, where meff = mAmB/(mA + mB).arrow_forwardThe CO molecule has a dipole moment of 0.480x10-30 C m. Given that its equilibrium bond distance is 212 pm, what is the magnitude of the effective charge that causes this dipole moment? Give your answer in units of the charge of an electronarrow_forwardSn, C, and Si, Ge are all group XIV elements. Yet, Sn is a conductor, C is an insulator while Si and Ge are semiconductors. Why?arrow_forward
- Although an ordinary H2 molecule consists of two identical atoms, this is not the case for the molecule HD, with one atom of deuterium (Le., heavy hydrogen, 2H). Because of its small moment of inertia, the HD molecule has a relatively large value of E: 0.0057 eV. At approximately what temperature would you expect the rotational heat capacity of a gas of HD molecules to "freeze out," that is, to fall significantly below the constant value predicted by the equipartition theorem?arrow_forwardThe atomic radii of a divalent cation and a monovalent anion are 0.074 nm and 0.128 nm, respectively. Calculate the force of attraction between these two ions at their equilibrium interionic separation (i.e., when the ions just touch one another) and the force of repulsion at the same distance.arrow_forwardA CO molecule is initially in the n = 2 vibrational level. If this molecule loses both vibrational and rotational energy and emits a photon, what are the photon wavelength and frequency if the initial angular momentum quantum number is l = 3?arrow_forward
 Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning

