
Concept explainers
Steam at 75 kPa and 8 percent quality is contained in a spring-loaded piston–cylinder device, as shown in Fig. P4–40, with an initial volume of 2 m3. Steam is now heated until its volume is 5 m3 and its pressure is 225 kPa. Determine the heat transferred to and the work produced by the steam during this process.
FIGURE P4–40
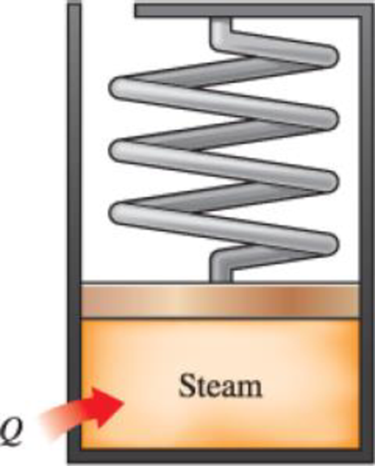
The heat transfer of the spring-loaded piston cylinder device.
The work done of the spring-loaded piston cylinder device.
Answer to Problem 40P
The heat transfer of the spring-loaded piston cylinder device is
The work done of the spring-loaded piston cylinder device is
Explanation of Solution
Write the expression for the energy balance equation.
Here, the total energy entering the system is
Substitute
Here, the mass of the piston cylinder device is
Calculate the specific volume of the spring-loaded piston cylinder device.
Here, the specific volume of saturated liquid is
Calculate the specific internal energy of the spring-loaded piston cylinder device.
Here, the specific internal energy of saturated liquid is
Write the expression for the mass of the system.
Here, the initial volume of the system is
Determine the final specific volume of the piston cylinder device.
The final volume of the piston cylinder device is
Determine the work done during the constant pressure process.
Here, the initial pressure is
Conclusion:
From the Table A-5, to obtain the value of the specific volume of saturated liquid is
Substitute 0.08 for
Substitute 0.08 for
Substitute
Substitute
From the Table A-5, to obtain the value of the specific volume of saturated liquid is
Determine the quality of final state for the spring-loaded piston-cylinder device.
Here, the specific volume of saturated liquid is
Substitute
Substitute 0.561375 for
Substitute
Thus, the heat transfer of the spring-loaded piston cylinder device is
Substitute
Thus, the work done of the spring-loaded piston cylinder device is
Want to see more full solutions like this?
Chapter 4 Solutions
CENGEL'S 9TH EDITION OF THERMODYNAMICS:
- Steam is contained in a 4-L volume at a pressure of 1.5 MPa and a temperature of 400 C. If the pressure is held constant by expanding the volume while 20 kJ of heat is added, the final temperature is nearestarrow_forwardConsider a closed piston cylinder that contains 0.2 kg of superheated water vapor at 200C and 100kPa. The cylinder is cooled and the piston moves is such a way that the pressure remains constant. At the end of the cooling process the water in the cylinder is a compressed liquid at 20C and 100kPa. Calculate the initial volume of the cylinder. Calculate the final volume of the cylinder. Calculate the work done on the system during the cooling process. How much heat was trasferred from the system to the surrounding area during the cooling process?arrow_forwardA cylinder having an initial volume of 3 m3 contains 0.1 kg of water at 40°C. The water is then compressed in an isothermal quasi-equilibrium process until it has a quality of 50%. Calculate the work done in the process. Assume the water vapor is an ideal gas.arrow_forward
- 0.2 m^3 of an ideal gas at a pressure of 2Mpa and 600 k is expanded isothermally to 5 times the initial volume. It is then cooled to 300 K at constant Volume and then compressed back polytropically to its initial state. Show the process on a P-V diagram and determine the work done.arrow_forward3-39 Water initially at 200 kPa and 300°C is contained in a piston-cylinder device fitted with stops. The water is allowed to cool at constant pressure until it exists as a saturated vapor and the piston rests on the stops. Then the water continues to cool until the pressure is 100 kPa. On the T-v diagram, sketch, with respect to the saturation lines, the process curves pass- 1stams! od on TURNACE ing through the initial, intermediate, and final states of the 1992 palam 11. water. Label the T, P, and U values for end states on the process artita ne odrow OFTİ 21 nucfortyd beinvarian curves. Find the overall change in internal energy between the 1610 PRICE initial and final states per unit mass of water. al state Onimals uliod Anub 1516w 3-150 3-160 In Water 200 kPa 300°C 3-17C ment rises. Now on top of air Obs will rise in a co FIGURE P3-39 180 Qarrow_forwardA frictionless piston–cylinder device, shown in initially contains 0.01 m3 of argon gas at 400 K and 350 kPa. Heat is now transferred to the argon from a furnace at 1200 K, and the argon expands isothermally until its volume is doubled. No heat transfer takes place between the argon and the surrounding atmospheric air, which is at 300 K and 100 kPa. Determine the maximum work that can be produced during this process.arrow_forward
- A cylinder fitted with a frictionless piston contains 5 kg of superheated vapor of ammonia at 1000 kPa and 140 C. The setup is cooled at constant pressure until the ammonia reaches a quality of 50%. Calculate the work done in the process and draw the process on T-v diagram.arrow_forward3–37E A spring-loaded piston-cylinder device is initially filled with 0.2 Ibm of an R-134a liquid-vapor mixture whose temperature is -30°F and whose quality is 80 percent. The spring constant in the spring force relation F = kx is 37 lbf/in, and the piston diameter is 12 in. The R-134a undergoes a process that increases its volume by 40 percent. Calculate the final temperature and enthalpy of the R-134a. Answers: 81.5°F, 120 Btu/lbm %3D Spring Fluid D- FIGURE P3-37Earrow_forwardThe spring-loaded piston–cylinder device shown in Fig. P3–63 is filled with 0.5 kg of water vapor that is initially at 4 MPa and 400°C. Initially, the spring exerts no force against the piston. The spring constant in the spring force relation F = kx is k = 0.9 kN/cm and the piston diameter is D = 20 cm. The water now undergoes a process until its volume is one-half of the original volume. Calculate the final temperature and the specific enthalpy of the water.arrow_forward
- Consider a piston-cylinder device that consists of 2 kg of R-134a at 40°C and x = 0.7. The system is cooled until the temperature drops to 18°C. State the final phase of R-134a in the piston cylinder device and give the volume (m³) of the system at final phase.arrow_forward(4) A mass of 3 kg of R-134a fill a piston-cylinder device with and initial volume of 0.279 m³ at a temperature of – 12.73°C. The refrigerant is now heated until the temperature is 40°C. The final volume of the R-134a is: (a) 0.322 m³ (b) 0.350 m³ (c) 0.385 m³ (d) 0.412 m³ (e) 0.446 m³arrow_forwardA piston-cylinder assembly initially contains 10 MPa pressure and 10 water vapor at 100 oC. Then, heating is carried out for 2 hours with the electric heater, which passes 220 V 20 A current at constant pressure to the water, during which time 7500 kJ of heat is transferred to the environment. Determine the final temperature of the water by showing this process on the T-v diagram.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





