
Concept explainers
Two tanks (Tank A and Tank B) are separated by a partition. Initially Tank A contains 2 kg of steam at 1 MPa and 300°C while Tank B contains 3 kg of saturated liquid–vapor mixture at 150°C with a vapor mass fraction of 50 percent. The partition is now removed and the two sides are allowed to mix until
FIGURE P4–44
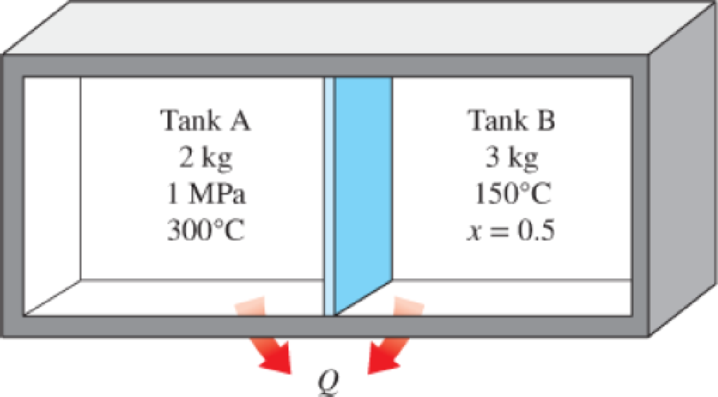
(a)
The final temperature of the steam for a tank.
The quality of the steam at the final state for a tank.
Answer to Problem 44P
The final temperature of the steam is
The quality of the steam at the final state is
Explanation of Solution
Write the expression for the energy balance equation.
Here, the total energy entering the system is
Simplify Equation (I) and write energy balance relation of two tanks.
Here, the boundary work to be done into the system is
Substitute 0 for
Here, the initial mass of tank A is
Write the expression for volume of tank A.
Here, the volume of the tank A is
Write the expression for volume of tank B.
Here, the volume of the tank B is
Write the expression for total mass of tank.
Write the expression for total volume contained in both tanks.
Write the expression for final specific volume of tank.
Conclusion:
Convert the unit of pressure from kPa to MPa.
From the Table A-6, “Superheated water”, obtain the value of initial specific volume
From the Table A-4, “Superheated water”, obtain the value of initial specific volume and specific internal energy for tank B.
At the initial temperature and quality of the steam at the final state of tank B as 150 C and 0.50.
Determine the specific volume of a two-phase system for tank B.
Here, the specific volume of saturated liquid for tank B is
Here, the specific internal energy of saturated liquid for tank B is
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Refer Table A-5, “Saturated water-pressure”, obtain the final temperature of the steam.
At final pressure of the steam is
Thus, the final temperature of the steam is
From the Table A-5, “Saturated water-pressure”, obtain the value of quality of the steam at the final state and final specific internal energy for tank B.
At the final pressure and specific volume at the final state as 300 kPa and
Determine the specific volume of a two-phase system for steam.
Here, the specific volume of saturated liquid for steam is
Here, the specific internal energy of saturated liquid for steam is
Substitute
Thus, the quality of the steam at the final state is
Substitute
(b)
The amount of heat lost from the tanks.
Answer to Problem 44P
The amount of heat lost from the tanks is
Explanation of Solution
Conclusion:
Substitute
Thus, the amount of heat lost from the tanks is
Want to see more full solutions like this?
Chapter 4 Solutions
THERMODYNAMICS (LL)-W/ACCESS >IP<
- Describe the equilibrium condition in terms of the entropy changes of a system and its surroundings. What does this de-scription mean about the entropy change of the universe?arrow_forwardRefrigerant-134a at 320 kPa and 40°C undergoes an isothermal process in a closed system until its quality is 45 percent. On a per-unit-mass basis, determine how much work and heat transfer are required.arrow_forwardTen grams of computer chips with a specific heat of 0.3 kJ/kg·K are initially at 20°C. These chips are cooled by placement in 5 grams of saturated liquid R-134a at –40°C. Presuming that the pressure remains constant while the chips are being cooled, determine the entropy change of the R-134a.arrow_forward
- A rigid and insulated tank of volume 2 m3is divided into two equal compartments by a partition. One compartment contains an ideal gas at 400 K and 3 MPa, while the other compartment contains the same gas at 600 K and 1 MPa. The partition is punctured and the gases are allowed to mix. Determine the entropy change of the gas. The isobaric molar heat capacity of the gas is equal to (5/2)arrow_forwardTen grams of computer chips with a specific heat of 0.3 kJ/kg·K are initially at 20°C. These chips are cooled by placement in 5 grams of saturated liquid R-134a at –40°C. Presuming that the pressure remains constant while the chips are being cooled, determine the entropy change of the entire system. Is this process possible? Why?arrow_forwardThe temperature of R-134a in a tank at a specified state is to be determined using the ideal gas relation, the van der Waals equation, and the refrigerant tables.arrow_forward
- Two rigid tanks are connected by a valve. Tank A contains 0.2 m3 of water at 400 kPa and 80 percent quality. Tank B contains 0.5 m3 of water at 200 kPa and 250°C. The valve is now opened, and the two tanks eventually come to the same state. Determine the pressure and the amount of heat transfer when the system reaches thermal equilibrium with the surroundings at 25°C.arrow_forwardDetermine the enthalpy of liquid water at 100°C and 15 MPa (a) by using compressed liquid tables, (b) by approximating it as a saturated liquid, and (c) by using the correction givenarrow_forwardOn a P-v diagram, what does the area under the process curve represent?arrow_forward
- A 50-kg copper block initially at 140°C is dropped into an insulated tank that contains 90 L of water at 10°C. Determine the final equilibrium temperature and the total entropy change for this processarrow_forwardWater is contained in a closed rigid tank at an initial pressure of 1200 kPa. Heat transfer occurs until the pressure increases to 7 MPa and the tank found to be containing 1,78 kg saturated liquid, and the mass of the saturated vapor is 0.22 kg. Determine the initial temperature, entropy and enthalpy of the water.arrow_forwardArgon gas enters an adiabatic compressor at 14 psia and 75°F with a velocity of 60 ft/s, and it exits at 200 psia and 240 ft/s. If the isentropic efficiency of the compressor is 87 percent, determine the exit temperature of the argon.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





