
Concept explainers
(a)
Interpretation:
The direction of the dipole in the following bond is to be determined.
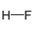
Concept Introduction:
The polar bond is formed between the two unlike atoms having electronegative difference ranges from 0.5-1.9. The polar bond represents the arrow towards the high electronegative element, while tail towards the low electronegative element. The negative sign is given to the high electronegative element while a positive sign is given to the low electronegative element.
(b)
Interpretation:
The direction of a dipole in the following bond is to be determined.
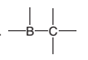
Concept Introduction:
The polar bond is formed between the two unlike atoms having electronegative difference ranges from 0.5-1.9. The polar bond represents the arrow towards the high electronegative element, while the tail towards the low electronegative element. The negative sign is given to the high electronegative element while a positive sign is given to the low electronegative element.
(c)
Interpretation:
The direction of a dipole in the following bond is to be determined.
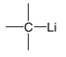
Concept Introduction:
The polar bond is formed between the two unlike atoms having electronegative difference ranges from 0.5-1.9. The polar bond represents the arrow towards the high electronegative element, while tail towards the low electronegative element. The negative sign is given to the high electronegative element while a positive sign is given to the low electronegative element.
(d)
Interpretation:
The direction of a dipole in the following bond is to be determined.
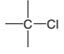
Concept Introduction:
The polar bond is formed between the two unlike atoms having electronegative difference ranges from 0.5-1.9. The polar bond represents the arrow towards the high electronegative element, while the tail towards the low electronegative element. The negative sign is given to the high electronegative element while a positive sign is given to the low electronegative element.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- For the bond between each of the following pairs of atoms, indicate the positive end with δ+ and the negative end with δ-. Draw an arrow to show the dipole for each. P and Cl Se and F N and H B and Clarrow_forwardAnswer the questions in the table below about the shape of the methanone (H₂CO) molecule. How many electron groups are around the central carbon atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central carbon atom? (You may need to use the scrollbar to see all the choices.) (choose one) X Sarrow_forwardArrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1 for the most polar bond, pick 2 for the next most polar bond, and so on. H H bond Se- —H -H HỘH polarity (Choose one) (Choose one) (Choose one) Xarrow_forward
- Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1 for the most polar bond, pick 2 for the next most polar bond, and so on. bond polarity H (Choose one) H H. H Н—с—н |(Choose one) H H-F: |(Choose one)arrow_forwardUse the symbols δ+ and δ- to indicate the polarity of the highlighted bonds.arrow_forwardBased on trends in electronegativity, Rank the following polarity of the bonds. Use numbers 1, 2, 3, 4 to fill in the ranking. 1 represents the MOST polar, 4 represents the LEAST polar! Molecule Н- СІ H - N Н-F Н-С H-CI [ Choose H-N [ Choose ] H-F [ Choose ] Н-С [ Choose ]arrow_forward
- Rank the elements or compounds in the table below in decreasing order of their boiling points. That is, choose 1 next to the substance with the highest boiling point, choose 2 next to the substance with the next highest boiling point, and so on. chemical symbol, substance chemical formula boiling point or Lewis structure A 0=N - Cl : (Choose one) В CaCl, (Choose one) v : Cl : : Cl: (Choose one) v :Cl : : Cl : Cl, (Choose one) ♥ ?arrow_forwardDraw the curved arrows on the left resonance structure to account for formation of the right resonance structure. DS NN S X 201 H C N 0 S F P Cl Br Iarrow_forwardHow do you know when to draw a solid wedge vs a dashed wedge when drawing 3D bond-line structures? I know that solid-wedge means the atom is pointing towards you and dashed wedge means it's in the back, but how do you know which atoms are in the front as opposed to the back? How can you tell what the configuration will look like in space just by looking at the lewis structure or name?arrow_forward
- Why do Period 2 elements never form more than four covalent bonds? Elements in Period 2: O have s, p and d subshells in their outer-most shell. O can hold at most 8 electrons in their outer shell. O can hold at most 18 electrons in their outer shell. O have only an s and p subshells in their outer-most shell. Why are Period 3 elements able to exceed an octet? Elements in Period 3: O can hold at most 8 electrons in their outer shell. O have s, p and d subshells in their outer-most shell. O have only ans and p subshells in their outer-most shell. O can hold at most 18 electrons in their outer shell. NOV tv 16 MacBook Airarrow_forwardDraw a Lewis structure for each ion.CH3O−HC2−(CH3NH3)+(CH3NH)−arrow_forwardDraw an equivalent resonance structure that minimizes charge. Include all lone pairs in your structure. CH2 0 Qarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





