
Concept explainers
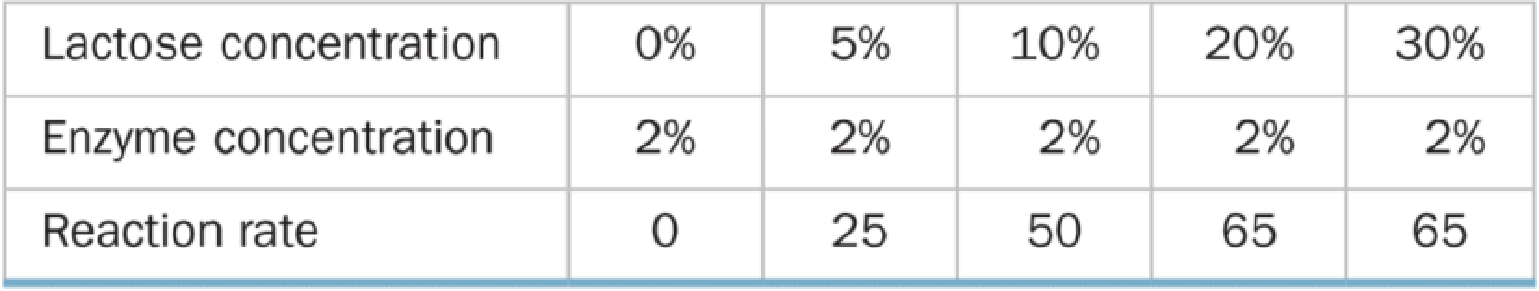
A biologist performed two series of experiments on lactase, the enzyme that hydrolyzes lactose to glucose and galactose. First, she made up 10% lactose solutions containing different concentrations of enzyme and measured the rate at which galactose was produced (grams of galactose per minute). Results of these experiments are shown in Table A below. In the second series of experiments (Table B), she prepared 2% enzyme solutions containing different concentrations of lactose and again measured the rate of galactose production.
Table A Reaction Rate and Enzyme Concentration
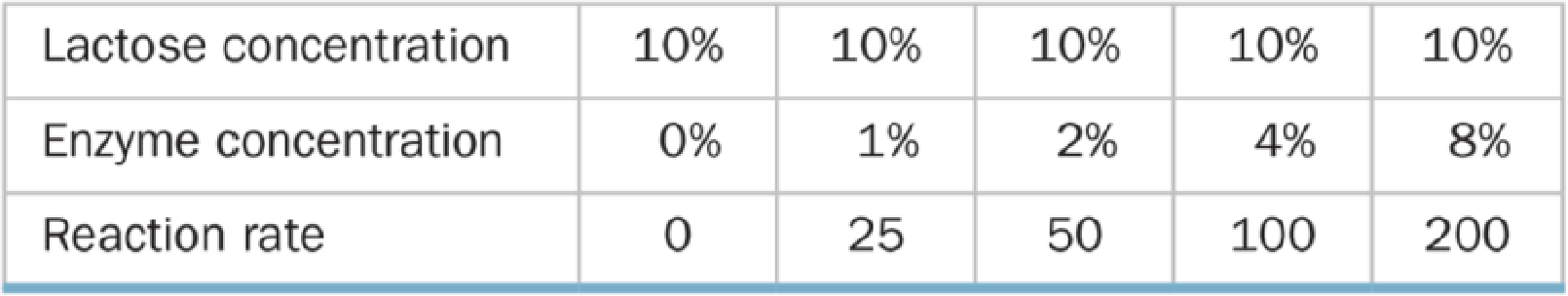
Table B Reaction Rate and Substrate Concentration
a. Graph and explain the relationship between the reaction rate and the enzyme concentration.
b. Graph and explain the relationship between the reaction rate and the substrate concentration. How and why did the results of the two experiments differ?
Trending nowThis is a popular solution!

Chapter 5 Solutions
Campbell Biology & Valuepack Access Card Pkg
- In an experiment where hydrogen peroxide is mixed with pieces of potato, which substance contains the enzyme?arrow_forwardThe purpose of this experiment is to determine how the activity of an enzyme can change under certain conditions: temperature, pH, substrate concentration Introduction Purpose statement is specific, complete and uses proper scientific vocabulary Hypothesis statement is clear, logical and scientifically sound Contains sufficient background information relevant to the lab Accurate use of examples and scientific terminology Clear, concise (max 1 page; single spaced) Step 1: Obtain a sample amount of a hard fruit or vegetable, such as an apple, cut it into smaller equal pieces, and measure on a digital scale of 10g. Step 2: Get 100ml of room temperature tap water and add it into a glass cup, heat it on a hot plate until reaches 37 Celsius Step 3: Add the apple sample into the glass cup with the tap water and mix gently. Let the mixture to sit for 5 minutes. Step 4: With a kitchen thermometer measure the temperature of the apple mixture. Step 5: In a glass cup add 3mL of 3% hydrogen…arrow_forwardAs you learned in class, many enzymes can catalyze the forward and reverse directions of a chemical reaction. If Lactaid was mixed with a solution that is very high in glucose and galactose, do you think Lactaid can catalyze the production of lactose? Please explain your answer in detail, and this question will require some additional research of your own.arrow_forward
- Leia wants to determine the effect of enzyme concentration on enzyme activity using potato samples and hydrogen peroxide. However, her results do not follow the theoretical trend and thus she can't draw any conclusions from the data obtained. What might have caused errors in the experiment? I. Placing potato samples under room temperature II. Different treatments, with potato being proportional to the volume of hydrogen peroxide III. Placed the same amount of hydrogen peroxide for a different amount of potatoes IV. Placed the same amount of potatoes for a different volume of hydrogen peroxidearrow_forwardWhat are the three kinds of enzyme-controlled reactions so that the chemical-bond energy from a certain nutrient is released to the cell in the form of ATP?arrow_forwardThe Enzyme Lactase Experiment Design How does pH affect how lactase drops work to break down lactose into glucose and galactose? What variable will you change in this experiment? This is also called the Independent variable. How will you change it? What variable will you measure in this experiment to determine the effect of the variable you changed? This is also called the Dependent variable. How will you measure it? What variables will you keep the same in this experiment? Check off the items in this list that you would use for this experiment. You may add any items you feel you would need. Skim milk (which contains lactose) Lactase drops Graduated cylinder Eye dropper Ice cubes pH paper Hot plate Mixing spoon Thermometer Vinegar (acidic) Test tubes Glucose test strips Baking soda (basic) Other items you feel you would need to use for this experiment. Describe what you would do in your experiment. For each step, explain why the step is important. Add…arrow_forward
- Which part of the curve shows the effect of an enzyme?arrow_forwardFor an experiment where you use different concentrations of catalase to determine how that affects rate of the reaction with its substrate, hydrogen peroxide, what effect does enzyme concentration have on reaction activity? Would it be possible to have too much catalase in the tube? What would happen?arrow_forwardThe normal enzyme required for converting sugars into glucose is present in cells, but the conversion never takes place and no glucose is produced. What could have occurred to cause this defect in a metabolic pathway?arrow_forward
- Which of the following is true about how enzymes work in the cell? A. Enzymes are consumed in the process of converting one molecule to another. B. Enzymes are needed to carry out only endergonic reactions. C. Enzymes are needed in the cell to increase the rate of a chemical reaction. D. Enzymes eliminate the activation barrier.arrow_forwardWhat is the mechanism that controls the rate of a reaction catalyzed by an enzyme?arrow_forwardIn an experiment where you use different concentrations of avocado catalase to determine how that affects rate of the reaction with its substrate, hydrogen peroxide, why was the amount(concentration) of peroxide kept the same for all tubes? Why was the total volume of every tube 10cm?arrow_forward
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
