
Concept explainers
Consider the following apparatus: a test tube coveredwith a nonpermeable elastic membrane inside a containerthat is closed with a cork. A syringe goes throughthe cork.
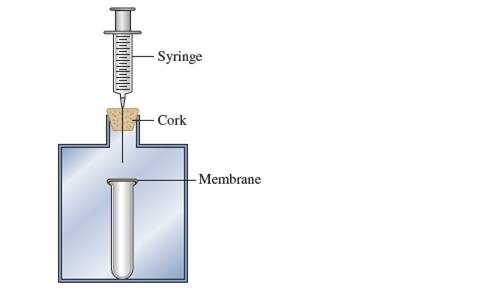
a. As you push down on the syringe, how does themembrane covering the test tube change?
b. You stop pushing the syringe but continue to hold itdown. In a few seconds, what happens to themembrane?
a)
Interpretation:Change in membrane cover if syringe is pushed down should be explained.
Concept introduction:Force per unit area is called pressure. Its SI unit is Pascal. Expression for pressure is as follows:
Explanation of Solution
When syringe is pushed down, pressure inside container and outside test tube increases. To balance effect of this increased pressure, elastic membrane expands in downward direction into test tube.
b)
Interpretation:Event that occurs if syringe is continued to be held down should be determined.
Concept introduction: Force per unit area is called pressure. Its SI unit is Pascal. Expression for pressure is as follows:
Explanation of Solution
When syringe is pushed down and hold down, constant pressure is applied on test tube and ultimately membrane will break.
Want to see more full solutions like this?
Chapter 5 Solutions
CHEM.PRINC.W/OWL2+REBATE+2 SUPPL.>IP<
- Why do liquids have a vapor pressure? Do all liquids have vapor pressures? Explain. Do solids exhibit vapor pressure? Explain. How does vapor pressure change with changing temperature? Explain.arrow_forward8.41 What is the specific feature of N, O, and F that causes them to play a role in hydrogen bonding?arrow_forwardA 0.500-g sample of zinc-copper alloy was treated with dilute hydrochloric acid. The hydrogen gas evolved was collected by water displacement at 27°C and a total pressure of 755 mm Hg. The volume of the water displaced by the gas is 105.7 mL. What is the percent composition, by mass, of the alloy? (Vapor pressure of H2O at 27°C is 26.74 mm Hg.) Assume only the zinc reacts.arrow_forward
- The shape of the meniscus of water in a glass tube is different from that of mercury in a glass tube. Why?arrow_forwardGive the formula of a solid containing Si that is (a) molecular (b) ionic (c) network covalentarrow_forwardYou and a friend each synthesize a compound with the formula XeCI2F2. Your compound is a liquid and your friend's compound is a gas (at the same conditions of temperature and pressure). Explain how the two compounds with the same formulas can exist in different phases at the same conditions of pressure and temperature.arrow_forward
- A plot of In (Pvap) versus 1/T (K) is linear with a negative slope. Why is this the case?arrow_forwardA common prank on college campuses is to switch the salt and sugar on dining hall tables, which is usually easy because the substances look so much alike. Yet, despite the similarity in their appearance, these two substances differ greatly in their properties, since one is a molecular solid and the other is an ionic solid. How do the properties differ and why?arrow_forwardConsider the iodine monochloride molecule, ICI. Because chlorine is more electronegative than iodine, this molecule is a dipole. How would you expect iodine monochloride molecules in the gaseous state to orient themselves with respect to each other as the sample is cooled and the molecules begin to aggregate? Sketch the orientation you would expect.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





