
Concept explainers
(a)
Interpretation: The stereogenic centers in the ball-and-stick model of ezetimibe are to be located.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
Answer to Problem 39P
The stereogenic centers in ezetimibe are,
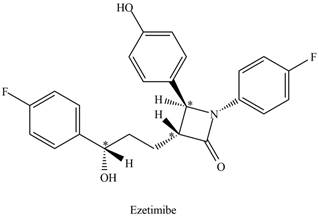
Explanation of Solution
The given ball-and-stick model of ezetimibe is shown below.
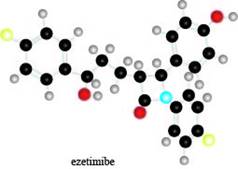
Figure 1
In the ball-and-stick model of ezetimibe, yellow balls represent halogen atom as they contain one covalent bond. Black balls are bonded to each other through four covalent bonds, thus; they represent carbon atom. Red balls represent oxygen atom as they contain two covalent bonds and blue balls represent nitrogen atom because they contain three covalent bonds.
The stereogenic center is one that has four different groups attached to carbon atom tetrahedrally. The stereogenic centers in the given molecule are located by omitting all
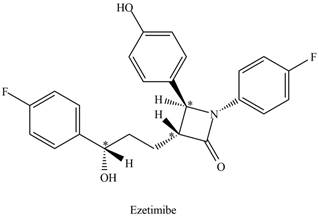
Figure 2
There are three stereogenic centers present in ezetimibe.
There are three stereogenic centers present in ezetimibe.
(b)
Interpretation: Each stereogenic center in ezetimibe are to be labeled as
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The naming of chiral center and geometric isomers are based on Cahn-Ingold-Prelog priority rules. If the priority assigned to each group attached to the chirality center in a molecule is in a clockwise direction, then it is the R-stereoisomer, and if this is counter-clockwise, then it is the S-stereoisomer. R and S-stereoisomer are mirror images of each other.
Answer to Problem 39P
The
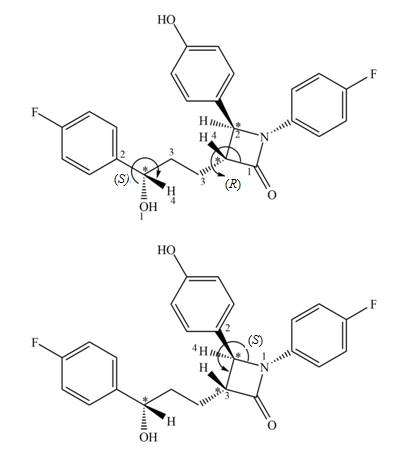
Explanation of Solution
The
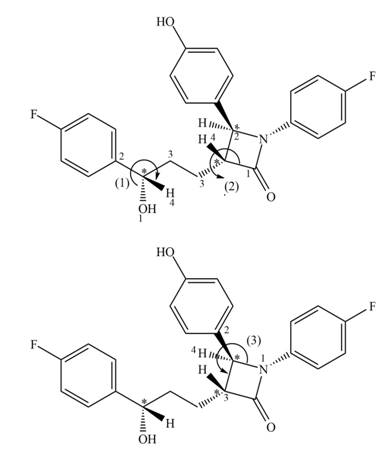
Figure 3
The stereogenic center (1) has
In ezetimibe, one of the stereogenic centers has
Want to see more full solutions like this?
Chapter 5 Solutions
ORGANIC CHEMISTRY-STUDY GDE./SOL.MAN.
- Captopril is a drug used to treat high blood pressure and congestiveheart failure.Draw the enantiomer of captoprilarrow_forwardCaptopril is a drug used to treat high blood pressure and congestiveheart failure.Designate each stereogenic center as R or S.arrow_forwarda.Label the four stereogenic centers in sorbitol as R or S. b.How are sorbitol and A related? c. How are sorbitol and B related?arrow_forward
- Locate the stereogenic centers in telaprevir, a drug used to treat hepatitis C, and label each stereogenic center as R or S.arrow_forwardDraw the structure of (S)-citalopram, a drug used to treat depression and anxiety that is much more potent than its R enantiomer.arrow_forwardLocate the stereogenic centers in each drug. Albuterol is a bronchodilator—that is, it widens airways—so it is used to treat asthma. Chloramphenicol isan antibiotic used extensively in developing countries because of its lowcost.arrow_forward
- Saquinavir (trade name Invirase) is a protease inhibitor, used to treat HIV (human immunodeficiency virus). a.Locate all stereogenic centers in saquinavir, and label each stereogenic center as R or S. b.Draw the enantiomer of saquinavir. c.Draw a diastereomer of saquinavir. d.Draw a constitutional isomer that contains at least one different functional group.arrow_forwardLabel the four stereogenic centers in sorbitol as R or S. (see attached file)arrow_forwardTrabectedin, shown in a ball-and-stick model on the cover of this text, isan anticancer drug sold under the trade name Yondelis. Question: Locate the stereogenic centers in trabectedin ?arrow_forward
- Hypoglycin A, an amino acid derivative found in unripened lychee, is a compound that is acutely toxic and can lead to death when ingested in large amounts by undernourished children. Draw all possible stereoisomers for hypoglycin A, and give the R,S designation for each stereogenic center.arrow_forwardHow is compound A related to compounds B–E? Choose from enantiomers, diastereomers, constitutional isomers, or identical molecules.arrow_forwardCelery ketone, like carvone, has two distinct aromas. In contrast to the S enantiomer's licorice scent, the R enantiomer has an earthy, celery-like aroma. Each enantiomer should be depicted and its odor assigned.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
