
(a)
Interpretation:
A correct set of four quantum numbers for each electron in boron atom has to be given.
Concept Introduction:
Principal Quantum Number (n): In an atom, the electron energy mainly depends on principal quantum number. The energy of an electron becomes lower when the value of n is smaller. The orbital size also depends on n. The size of orbital increases with increase in value of principal quantum number (n)
Magnetic Quantum Number (
Spin Quantum Number (
(a)
Explanation of Solution
The electronic configuration of boron atom is given by
The arrangement of electrons in boron atom is
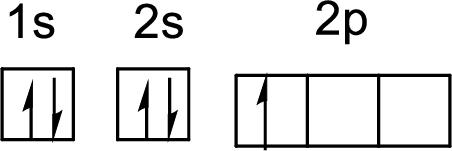
First electron in 1s orbital:
Second electron in 2s orbital:
First electron in 2s orbital:
Second electron in 2s orbital:
First electron in 2p orbital:
(b)
Interpretation:
A correct set of four quantum numbers for
Concept Introduction:
Refer to part (a)
(b)
Explanation of Solution
The electronic configuration of magnesium atom is given by
The arrangement of

First electron in
Second electron in
(c)
Interpretation:
A correct set of four quantum numbers for a
Concept Introduction:
Refer to part (a)
(c)
Explanation of Solution
The electronic configuration of iron atom is given by
The arrangement of a
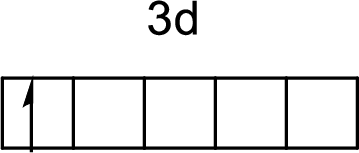
First electron in
Want to see more full solutions like this?
Chapter 5 Solutions
EBK CHEMISTRY: THE MOLECULAR SCIENCE
- What experimental evidence supports the quantum theory of light? Explain the wave-particle duality of all matter .. For what size particles must one consider both the wave and the particle properties?arrow_forwardA bright violet line occurs at 435.8 nm in the emission spectrum of mercury vapor. What amount of energy, in joules, must be released by an electron in a mercury atom to produce a photon of this light?arrow_forwardThe cesium atom has the lowest ionization energy, 375.7kJmol1 , of all the neutral atoms in the periodic table. What is the longest wavelength of light that could ionize a cesium atom? In which region of the electromagnetic spectrum does this light fall?arrow_forward
- Potassium atoms in a flame emit light as they undergo transitions from one energy level to another that is 4.91019J lower in energy. Calculate the wavelength of light emitted and, by referring to Figure 4.3, predict the color visible in the flame.arrow_forwardImagine a world in which the rule for the l quantum number is that values start with 1 and go up to n. The rules for the n and mi quantum numbers are unchanged from those of our world. Write the quantum numbers for the first two shells (i.e., n = 1 and n = 2).arrow_forward6.96 When a helium atom absorbs light at 58.44 nm, an electron is promoted from the 1s orbital to a 2p orbital. Given that the ionization energy of (ground state) helium is 2372 kJ/ mol, find the longest wavelength of light that could eject an electron from the excited state helium atom.arrow_forward
- Use the mathematical expression for the 2pz wave function of a one-electron atom (see Table 5.2) to show that the probability of finding an electron in that orbital anywhere in the x-y plane is 0. What are the nodal planes for a dxz orbital and for a dx2y2 orbital?arrow_forward6.32 What are the mathematical origins of quantum numbers?arrow_forwardCalculate the wavelength of the Balmer line of the hydrogen spectrum in which the initial n quantum number is 5 and the final n quantum number is 2.arrow_forward
- What type of electron orbital (i.e., s, p, d, or f) is designated by an electron with quantum numbers (a) n=1,l=0,m l =0(b) n=3,l=2,m l =1? (c) n=4,l=3,m l =3arrow_forwardWhich of the following sets of quantum numbers correctly represents a 4p orbital? (a) n = 4, = 0, m = 1 (b) n = 4, = 1, m = 0 (c) n = 4, = 2, m = 1 (d) n = 4, = 1, m =2arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





