
(a)
Interpretation:
The temperature dependence of the vapor pressure of phosphoryl chloride difluoride is given, ln p against 1/T has to be plotted.
Concept introduction:
The relationship to plot is In P versus 1/T is given below.
(a)
Explanation of Solution
Given,
| Temperature, T/K | 190 | 228 | 250 | 273 |
| Vapour pressure, P/Torr | 3.2 | 68 | 240 | 672 |
Unit conversion is given below,
Therefore,
| Temp. | V.P (Torr) | V.P (atm) | lnP | |
The ln p against 1/T plot is given below,
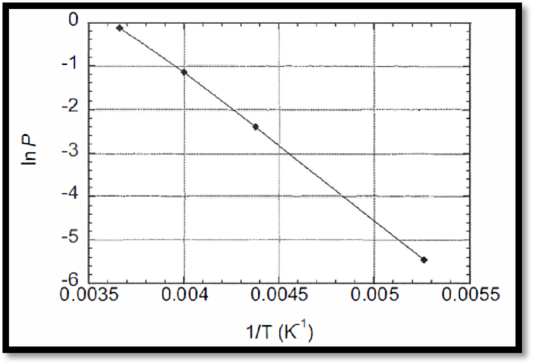
Figure 1
From the curve, slope is given below,
(b)
Interpretation:
The enthalpy of the vaporization has to be calculated.
Concept introduction:
Refer to part (a)
(b)
Explanation of Solution
The relationship to plot is In P versus 1/T, this gives a straight line.
From the curve, slope is given below,
The enthalpy of the vaporization is
(c)
Interpretation:
The entropy of the vaporization has to be calculated.
Concept introduction:
Refer to part (a)
(c)
Explanation of Solution
The relationship to plot is In P versus 1/T, this gives a straight line.
From the curve, slope is given below,
The entropy of the vaporization is
(d)
Interpretation:
The normal boiling point of phosphoryl chloride difluoride has to be calculated.
Concept introduction:
Refer to part (a)
(d)
Explanation of Solution
The normal boiling point of phosphoryl chloride difluoride is calculated when the vapor pressure is
Therefore, it will happen when.
The normal boiling point of phosphoryl chloride difluoride is
(e)
Interpretation:
If the pressure is 15 torr, temperature of phosphoryl chloride difluoride has to be calculated.
Concept introduction:
Refer to part (a)
(e)
Explanation of Solution
The temperature of phosphoryl chloride difluoride value is calculated as follows,
The temperature of phosphoryl chloride difluoride value is
Want to see more full solutions like this?
Chapter 5 Solutions
EBK CHEMICAL PRINCIPLES
- Calculate the enthalpy of solution for the dissolution of lithium iodide, LiI, molar mass = 133.85 g mol-1. When 1.49 g of LiI is dissolved in a coffee cup calorimeter containing 75.0 mL of water the temperature increased from 23.5 °C to 25.7 °C. The specific heat of water is 4.184 J g-1 °C-1, and assume the density of the solution is 1.00 g mL-1. -62 kJ mol-1 -39 kJ mol-1 30 kJ mol-1 -76 kJ mol-1 18 kJ mol-1arrow_forwardEstimate the temperature at which the equilibrium constant for the decomposition of CaCO3(s, calcite) to CO2(g) and CaO(s) becomes 1; assume pCO2 = 1 bar .arrow_forwardCertain bacteria in the soil obtain the necessary energy for growth by oxidizing nitrite to r nitrate: 2NO2- (aq) + O2(g) —> 2NO3-(aq) Given that the standard Gibbs energies of formation of NO2- and NO3- are -34.6 kJ mol-1 and -110.5 kJ mol-1, respectively, calculate the amount of Gibbs energy released when 1 mole of No2- is oxidized to 1 mole of NO3-.arrow_forward
- What partial pressure of hydrogen results in a molar concentration of 1.0 mmol dm-3 in water at 25 °c?arrow_forwardThe standard Gibbs energy of formation of rhombic sulfur is zero and that of monoclinic sulfur is +0.33 kJ mol-1, at 25 °c.Which polymorph is the more stable at that temperature?arrow_forwardThe osmotic pressure at the base of a houseplant kept at 27ºC is 1.6 × 104 N/m2. Calculate the molar concentration of the dissolved molecules in the plant’s veins if there are no dissolved molecules in the soil water.arrow_forward
- Calculate (a) the standard reaction entropy and (b) the change in entropyof the surroundings (at 298 K) of the reaction N2(g) + 3 H2(g) → 2 NH3(g). (c) Hence calculate the standard Gibbs energy of the reaction.arrow_forwardIf the equilibrium constant of a reaction is Keq = 9.3 × 103 at 900 °C, what is the change in Gibbs free energy for this reaction (in kJ mol–1) at the indicated temperature? R = 8.314 J K–1 mol–1arrow_forwardThe standard Gibbs free...arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





