
Concept explainers
The following four structures are naturally occurring optically active compounds. Star (*) the asymmetric carbon atoms in these structures.
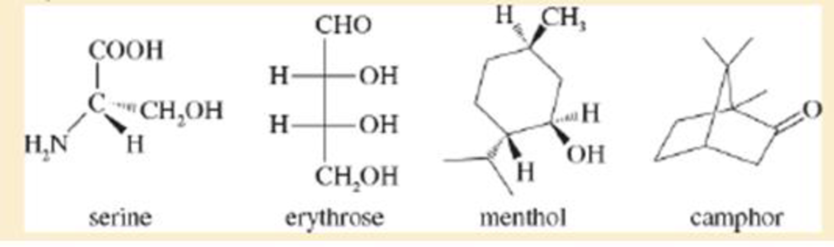
Interpretation: The asymmetric carbon atoms in the given structures are to be marked by star.
Concept introduction: A chiral carbon atom is attached to four different atoms or group of atoms and shows a tetrahedral geometry. The mirror image of a chiral compound is non-super imposable. The two different forms in which a single chiral carbon can exist are referred as enantiomers. The number of enantiomers of a molecule depends on the number of chiral centers.
To determine: The asymmetric carbon atoms in the given structures marked by star.
Answer to Problem 5.25SP
The asymmetric carbon atoms in the given structure have been marked by star.
Explanation of Solution
The given compound is serine. It is attached to
The asymmetric carbon atom present in it is marked by star as shown below.
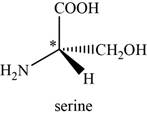
Figure 1
The given compound is erythrose. There are two chiral centres present in it.
The asymmetric carbon atoms present in it are marked by star as shown below.
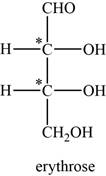
Figure 2
The given compound is menthol. There are three chiral centres present in it.
The asymmetric carbon atoms present in it are marked by star as shown below.
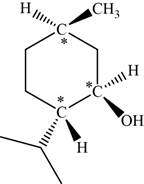
Figure 3
The given compound is camphor. There are two chiral centres present in it.
The asymmetric carbon atoms present in it are marked by star as shown below.
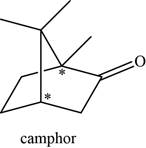
Figure 4
The asymmetric carbon atoms in the given structure have been marked by star.
Want to see more full solutions like this?
Chapter 5 Solutions
Student's Solutions Manual for Organic Chemistry
Additional Science Textbook Solutions
Chemistry For Changing Times (14th Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry (7th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry In Context
General Chemistry: Principles and Modern Applications (11th Edition)
- Quiana whose structure is shown below is a synthetic fabric that feels very much like silk. What are the structures of the two monomers that are used to make Quiana?arrow_forwardA student drew a noncyclic compound that has the molecular formula C7H14.This molecular formula is representative of an alkane, alkene, alkyne or aromatic and would be classified as saturated or unsaturated ?arrow_forwardChemistry Circle the chiral carbons in the following compounds.arrow_forward
- Circle all of the chiral centers (i.e., asymmetric carbon atoms) in the following compounds.arrow_forwardwhat is a stereoisomer of the molecule trans-1,2-Dichloroethene?arrow_forwardOrganic Chemistry HW: CANNOT BE HAND DRAWN 2,6-dimethyloct-2-ene Provide a detailed typed explanation of Stereoisomers show the expanded structure of your molecule. Calculate the maximum number of possible stereoisomers of your molecule using the following formula: Maximum number of possible stereoisomers = 2n (where n= the number of chiral carbons in your molecule). This calculation does not include E- or Z- isomers for any compounds containing double bonds Type or using a computer program "draw" the possible stereoisomers of the molecule. Note that E-, Z- isomers of each stereoisomer are also possible and would not be accounted for by the formula above; draw any E- or Z- isomers.arrow_forward
- A. Which class/classes* of hydrocarbons is/are reactive to Br2 in CH2Cl2 ONLY in the presence of light? b. Which class/classes of hydrocarbons is/are reactive to Br2 in CH2Cl2 WITH OR WITHOUT light? c. Which class/classes of hydrocarbons is/are not reactive to Br2 in CH2Cl2 BOTH in the presence and absence of light?arrow_forwardIllustrate the Biologically active organic compounds that contain conjugateddouble bonds ?arrow_forwardHow many chiral centers are found in this formula?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

