
Concept explainers
(a)
Interpretation: The gas with highest partial pressure in the mixture of four gases; A (purple), B (black), C (green) and D2(orange) needs to be determined.
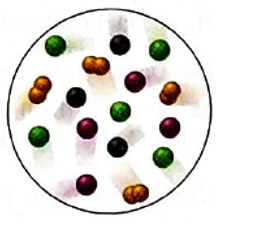
Concept Introduction:According to Dalton’s law of partial pressure, the total pressure of a gaseous mixture is equal to the sum of partial pressures of all gaseous components. Hence, the total pressure is larger than the partial pressure of both gaseous components.
Mole fraction is the ratio of moles of substance and total moles in the solution or mixture.
(b)
Interpretation: The gas with the lowest partial pressure in the mixture of four gases; A (purple), B (black), C (green) and D2(orange) needs to be determined.
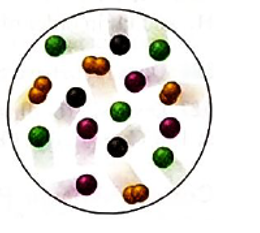
Concept Introduction: According to Dalton’s law of partial pressure, the total pressure of a gaseous mixture is equal to the sum of partial pressures of all gaseous components. Hence the total pressure is larger than the partial pressure of both gaseous components.
Mole fraction is the ratio of moles of substance and total moles in the solution or mixture.
(c)
Interpretation: The partial pressure of D2 gas needs to be determined if the total pressure if the gas mixture is 0.75 atm in the mixture of four gases; A (purple), B (black), C (green) and D2(orange).
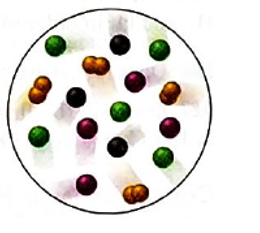
Concept Introduction: According to Dalton’s law of partial pressure, the total pressure of a gaseous mixture is equal to the sum of partial pressures of all gaseous components. Hence, the total pressure is larger than the partial pressure of both gaseous components.
Mole fraction is the ratio of moles of substance and total moles in the solution or mixture.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Principles of General Chemistry
- A collapsed balloon is filled with He to a volume of 12.5 L at a pressure of 1.00 atm. Oxygen, O2, is then added so that the final volume of the balloon is 26 L with a total pressure of 1.00 atm. The temperature, which remains constant throughout, is 21.5 C. (a) What mass of He does the balloon contain? (b) What is the final partial pressure of He in the balloon? (c) What is the partial pressure of O2 in the balloon? (d) What is the mole fraction of each gas?arrow_forwardIf equal masses of O2 and N2 are placed in separate containers of equal volume at the same temperature, which of the following statements is true? If false, explain why it is false. (a) The pressure in the flask containing N2 is greater than that in the flask containing O2. (b) There are more molecules in the flask containing O2 than in the flask containing N2.arrow_forwardButyl mercaptan, C4H9SH, has a very bad odor and is among the compounds added to natural gas to help detect a leak of otherwise odorless natural gas. In an experiment, you burn 95.0 mg of C4H9SH and collect the product gases (SO2, CO2, and H2O) in a 5.25-L flask at 25 C. What is the total gas pressure in the flask, and what is the partial pressure of each of the product gases?arrow_forward
- Hydrogen gas is used in weather balloon because it is less expensive than Helium. Assume that 5.57 g of H2 is used to fill a weather balloon to an initial volume of 67 L at 1.04 atm. If the ballloon rises to an altitude where the pressure is 0.047 atm, what is its new volume? Assume that the temperature remains constant.arrow_forwardShown below are three containers of an ideal gas (A, B, and C), each equipped with a movable piston (assume that atmospheric pressure is 1.0 atm). a How do the pressures in these containers compare? b Are all the gases at the same temperature? If not, compare the temperatures. c If you cooled each of the containers in an ice-water bath to 0.0C, describe how the volumes and pressures of the gases in these containers would compare.arrow_forward47 HCl(g) reacts with ammonia gas, NH3(g), to form solid ammonium chloride. If a sample of ammonia occupying 250 mL at 21 C and a pressure of 140 torr is allowed to react with excess HCl, what mass of NH4Cl will form?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





