
Interpretation:
Chiral carbon atoms present in nepetalactone has to be labelled. The number of possible stereoisomers for nepetalactone has to be given and to show that nepetalactone is a terpene.
Concept Introduction:
Terpenes are made by joining five-carbon units, usually in a head to tail-fashion.
Isoprene unit:
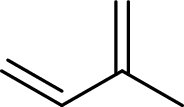
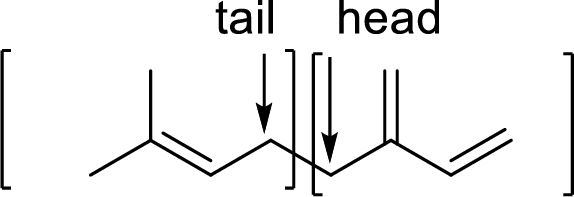
Branched end of isoprene – Head
Unbranched end of isoprene - Tail
Chirality: It refers to a Carbon atom in a molecule that contains four different substituents.
Stereoisomers: Two compounds with same molecular formula but different in their orientation are considered as isomers.
Number of possible stereoisomers for a compound can be determined as,
Trending nowThis is a popular solution!

Chapter 5 Solutions
Student Study Guide and Solutions Manual for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- What type of stereochemistry, S or R, does the chiral center in this molecule have?arrow_forwardConstruct models of all the possible stereoisomers of 1,2-dichlorocyclopropane. Using your models, draw structures for and name the isomers. Label each chirality centre R or S. Indicate which pairs are related as enantiomers and which as diastereoisomers.arrow_forwardThe molecule 2,5-dibromohex-3-ene contains three stereogenic sites: a double bond that can be cis- or trans- and two chirality centers that can be either (R) or (S). Draw structures for and name all possible stereoisomers of 2,5-dibromohex-3-ene. HINT: That means a possible total of 2³ stereoisomers, but there's also the potential for high symmetry or meso stereoisomers, which then reduces the total number of possibilities. Put boxes around pairs of enantiomers, and label meso compounds as "meso," as appropriate.arrow_forward
- Draw and name all possible isomers of trimethylcyclopropane. Which of these isomers are chiral?arrow_forwardassign the stereochemical configuration of the selected tetrahedral carbon chiral centers (R, S or N (not a chiral center)) and the alkene (E, Z or N (not a stereocenter)) that are indicated by the arrows (note that you do not have to assign the configuration of every chiral center in the molecule). If the atom in question is not a chiral center or is not a stereocenter circle N for neither.arrow_forward5. For the molecules below assign the stereochemical configuration of the selected tetrahedral carbon chiral centers (R, S or N (not a chiral center)) and the alkene (E, Z or N (not a stereocenter)) that are indicated by the arrows (note that you do not have to assign the configuration of every chiral center in the molecule). If the atom in question is not a chiral center or is not a stereocenter circle N for neither. b) RSN RSN HO" RSN EZ N RSN НО. H₂N HO HO OH HO OH RSN RSN CH3 НИ H3 CH₂ RSN RSN RSN EZ N RSNarrow_forward
- Stereoisomers share the same connectivity and differ only in the way their atoms are arranged in space. Draw the structure of a compound that is a stereoisomer of cis-1,2-dimethylcyclobutane.(Note that the question asks for a different stereoisomer of the named compound and not the named compound itself.)Use the wedge/hash bond tools to indicate stereochemistry where it exists.In cases where there is more than one answer, just draw one.arrow_forward(a) Describe the molecular geometry expected for 1,2,3-butatriene (H2C=C=C=CH2). (b) Two stereoisomers are expected for 2,3,4-hexatriene (CH3CH=C=C=CHCH3). What should be the relationship between these two stereoisomers?arrow_forward2. Explain with a couple of acyclic or cyclic examples (5 carbons or more) what is crit- ical for you when establishing whether a molecule is chiral or acral.arrow_forward
- Pyrethrins, such as jasmolin II(below), are a group of naturalcompounds synthesized by flowers of the genus Chrysanthemum(known as pyrethrum flowers) to act as insecticides.(a) Circle and name the functional groups in jasmolin II.(b) What is the hybridization of the numbered carbons?(c) Which, if any, of the numbered carbons are chiral centers?arrow_forwardIs pentanoic acid a chiral molecule or not?arrow_forwardSquare planar molecules with formula AB2C2 and octahedral molecules with formulas AB4C2 and AB3C3 feature diastereoisomers. Recall that trigonal bipyramidal geometry features two distinct positions: axial and equatorial. Draw all diastereoisomers for trigonal bipyramidal molecules with formula (a) AB4C and (b) AB3C2. You must indicate the stereochemistry using full and dashed wedges and label all positions as either axial (ax) and equatorial (eq)..arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

