
GENERAL ORGANIC+BIOL...-MASTERINGCHEM.
4th Edition
ISBN: 9780135170748
Author: FROST
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.38PP
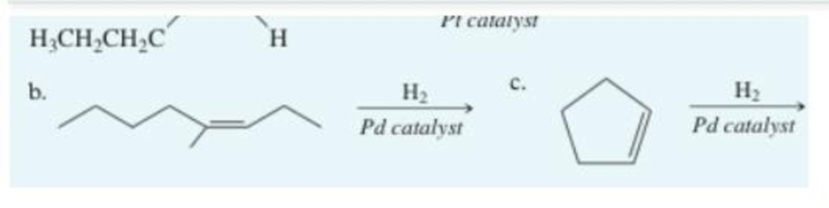
Write the main product of hydration for the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. When, a volume of 200 cm³ of Lithium hydroxide solution of concentration 1.00 mol/dm³
and 200 cm³ of Hydrochloric acid of concentration 100 mol/dm³ were mixed together in a
calorimeter the reaction was investigated to be exothermic.
a) Distinguish between endothermic and exothermic reactions.
b) Based on the information given above in question one (1), calculated the enthalpy
change per mol of water produced, if the heat capacity of the calorimeter was 76 J/K
and the temperature rise was 5.25 K. (State any assumptions made)
True or false: A reaction that has a Gibbs free energy of -3 kcal/mol is an endergonic (non-spontaneous) reaction.
True
False
In liters, how much oxygen and STP are required to react with excess methane within this following reaction, in order to produce 325 kJ of energy?
CH4 + 2 O2 -> CO2 + H2O
ΔH = -890.4 kJ
Chapter 5 Solutions
GENERAL ORGANIC+BIOL...-MASTERINGCHEM.
Ch. 5 - When vinegar (CH3COOH) and baking soda (NaHCO3)...Ch. 5 - In your own words, define free energy change, G....Ch. 5 - Classify the following as exothermic or...Ch. 5 - Prob. 5.4PPCh. 5 - Prob. 5.5PPCh. 5 - Classify the following as spontaneous or...Ch. 5 - Prob. 5.7PPCh. 5 - Prob. 5.8PPCh. 5 - Prob. 5.9PPCh. 5 - Prob. 5.10PP
Ch. 5 - a. How does increasing the temperature increase...Ch. 5 - a. Describe activation energy for a chemical...Ch. 5 - Why does the rate of a chemical reaction decrease...Ch. 5 - Prob. 5.14PPCh. 5 - Enzymes increase the rate of a biological chemical...Ch. 5 - Prob. 5.16PPCh. 5 - Prob. 5.17PPCh. 5 - Prob. 5.18PPCh. 5 - Categorize the following reactions as synthesis,...Ch. 5 - Categorize the following reactions as synthesis,...Ch. 5 - Prob. 5.21PPCh. 5 - Prob. 5.22PPCh. 5 - Write the products and balance the following...Ch. 5 - Prob. 5.24PPCh. 5 - Prob. 5.25PPCh. 5 - List the differences between general chemical...Ch. 5 - Are the substances shown in italics undergoing...Ch. 5 - Prob. 5.28PPCh. 5 - Prob. 5.29PPCh. 5 - Prob. 5.30PPCh. 5 - Prob. 5.31PPCh. 5 - Prob. 5.32PPCh. 5 - Prob. 5.33PPCh. 5 - Prob. 5.34PPCh. 5 - Prob. 5.35PPCh. 5 - Prob. 5.36PPCh. 5 - Prob. 5.37PPCh. 5 - Write the main product of hydration for the...Ch. 5 - Methane (a.k.a. natural gas) can react with oxygen...Ch. 5 - Prob. 5.40APCh. 5 - Which reaction occurs at a faster rate, an...Ch. 5 - Prob. 5.42APCh. 5 - Prob. 5.43APCh. 5 - Two curves for the same reaction are shown in the...Ch. 5 - Prob. 5.45APCh. 5 - Draw and label a reaction energy diagram for an...Ch. 5 - Prob. 5.47APCh. 5 - Prob. 5.48APCh. 5 - Write the products that would result from the...Ch. 5 - Prob. 5.50APCh. 5 - Prob. 5.51APCh. 5 - Prob. 5.52APCh. 5 - Prob. 5.53APCh. 5 - Prob. 5.54APCh. 5 - Prob. 5.55APCh. 5 - Identify the reactant that is oxidized and the...Ch. 5 - Prob. 5.57APCh. 5 - Write the products of the following reactions:Ch. 5 - Prob. 5.59APCh. 5 - Acetylsalicylic acid (aspirin) can be synthesized...Ch. 5 - Prob. 5.61APCh. 5 - Fill in the missing organic produce for the...Ch. 5 - Prob. 5.63APCh. 5 - Prob. 5.64APCh. 5 - How do low-carb diets work? We store glucose...Ch. 5 - Prob. 5.66CPCh. 5 - Prob. 5.67CPCh. 5 - Prob. 5.68CPCh. 5 - Prob. 5.69CPCh. 5 - Which reaction has the larger activation energy?Ch. 5 - Prob. 1IA.2QCh. 5 - A catalyst speeds up a chemical reaction by...Ch. 5 - Prob. 1IA.4QCh. 5 - Examine your sketch from question 3. Does a...Ch. 5 - Prob. 1IA.6QCh. 5 - Prob. 2IA.1QCh. 5 - Prob. 2IA.2QCh. 5 - Prob. 2IA.3QCh. 5 - One of the reactions in the data set is a single...Ch. 5 - Prob. 2IA.5QCh. 5 - Categorize the following reactions as a synthesis,...Ch. 5 - Prob. 3IA.1QCh. 5 - Prob. 3IA.2QCh. 5 - Prob. 3IA.3QCh. 5 - Prob. 3IA.4QCh. 5 - Prob. 3IA.5QCh. 5 - Prob. 1ICCh. 5 - Find out how unsaturated fats are saturated and...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Would the amount of heat absorbed by the dissolution in Example 5.6 appear greater, lesser, or remain the same if the experimenter used a calorimeter that was a poorer insulator than a coffee cup calorimeter? Explain your answer.arrow_forwardAlthough the gas used in an oxyacetylene torch (Figure 5.7) is essentially pure acetylene, the heat produced by combustion of one mole of acetylene in such a torch is likely not equal to the enthalpy of combustion of acetylene listed in Table 5.2. Considering the conditions for which the tabulated data are reported, suggest an explanation.arrow_forwardb) The standard enthalpy change of combustion (i.e. the heat change) of sucrose is -5644 kJ mol-¹. Calculate the energy released when one sweet containing 6.70 g of sucrose is completely burnt. c) A man needs to consume about 2500 dietary calories (i.e. 2500 kcal) per day. Given that 1 kJ = 0.239 kcal, how many sweets would a man have to consume in order to meet his daily calorific requirement? Sherbet produces a fizzing sensation in the mouth when its two main components, tartaric acid and sodium hydrogencarbonate, react together in aqueous solution to make carbon dioxide gas. The reaction is given below: H₂C₂H4O6 + 2NaHCO3 Sodium Bicarbonate Tartaric Acid Na₂C4H₂O6 + 2H₂O + Sodium Water Tartarate 2CO₂ Carbon Dioxidearrow_forward
- 2. A 1.07 g sample of sulfur was burned in a bomb calorimeter with calorimeter constant of 2.03 kcal/°C. If the resulting temperature change was 1.17°C, what is the molar heat of combustion for sulfur in kcal/mole? 1.11arrow_forwardAmmonification is the process by which A B C ammonia is released during the decomposition of nitrogen-containing organic compounds ammonium is converted to nitrite and nitrate in soils nitrate from soil is transformed to gaseous nitrogen compounds such as NO, N₂O, and N₂ D gaseous nitrogen is fixed to yield ammonia 4arrow_forwardAspirin can be made in the laboratory by reacting acetic anhydride (C4H6O3) with salicylic acid (C7H6O3) to form aspirin (C9H8O4) and acetic acid (C2H4O2). The balanced equation is:C4H6O3+C7H6O3→C9H8O4+C2H4O2.In a laboratory synthesis, a student begins with 5.00 mL of acetic anhydride (density = 1.08 g / mL) and 2.08 g of salicylic acid. Once the reaction is complete, the student collects 2.17 g of aspirin. Determine the limiting reactant for the reaction. Express your answer as a chemical formula.arrow_forward
- Aspirin can be made in the laboratory by reacting acetic anhydride (C4H6O3) with salicylic acid (C7H6O3) to form aspirin (C9H8O4) and acetic acid (C2H4O2). The balanced equation is:C4H6O3+C7H6O3→C9H8O4+C2H4O2.In a laboratory synthesis, a student begins with 5.00 mL of acetic anhydride (density = 1.08 g / mL) and 2.08 g of salicylic acid. Once the reaction is complete, the student collects 2.14 g of aspirin. -Determine the limiting reactant for the reaction.Express your answer as a chemical formula. -Determine the theoretical yield of aspirin for the reaction. -arrow_forwardHow HgSO4 is used ?arrow_forwardThe complete combustion of 1.5 moles ofmethane (CH4) would require how much O2?arrow_forward
- During the combustion of 5.00 gg of octane, C8H18C8H18, 239.5 kcalkcal (1002 kJkJ) is released. what is the balanced equation for the combustion reaction? How much energy (in kJkJ) is released by the combustion of 1.21 molmol of C8H18C8H18 ? How many moles of octane must be burned to release 442.4 kcalkcal ? How many kilocalories are released by the combustion of 18.3 gg of C8H18C8H18 ?arrow_forwardThe complete combustion of propane gas can be represented as a. 2 C3H8 + 7 02→ 2 C+ 2 CO2 + 8 H2O + heat b. 2 C3Hs + 7 02→ 2 C + 2 CO + 2 CO2 + 8 H2O + heat c. C3H8 + 10 02→3 CO2+ 4 H20 + heat d. C3Hg + 7 02 + CO2 → 4 CO2 + 4 H20 + heatarrow_forwardHow much oxygen is needed to produce 105.2 kJ via the following reaction?2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(l) ΔH=-3120 kJ/molMolar masses: C2H6: 30.07 g/mol; O2: 32.00 g/mol; CO2: 44.01 g/mol; H2O: 18.02 g/molarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY