
Concept explainers
(a)
Interpretation:
Whether the compounds in the given pair have the same or different boiling points is to be determined.
Concept introduction:
Isomers are the pair of compounds that have the same formula.
Constitutional isomers are the isomers having the same molecular formula but different connectivity. Constitutional isomers must have different physical and chemical properties.
The configurational isomers are not interconvertible by rotating around a single bond.
Enantiomers are configurational isomers having the same connectivity but cannot be interconvertible by rotation around a single bond. The mirror images of enantiomers are non-superimposable. Since enantiomers have the same connectivity of atoms, they should behave identically.
Diastereomers are configurational isomers having same connectivity but are not mirror images of each other. Diastereomers show different physical and chemical properties. Cis-trans isomers are diastereomers.
The boiling point of isomers comes under physical property.
Answer to Problem 5.53P
The given pair of isomers have different boiling points as they are diastereomers of each other.
Explanation of Solution
The given pair of compounds is
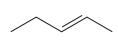
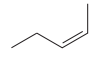
Both the molecules in the given pair have the same molecular formula; they also have the same connectivity. Thus, they are configurational isomers. The given pair of molecules is cis-trans isomers. So, these compounds are diastereomers of each other. Diastereomers show different physical and chemical properties. Thus, the compounds in the given pair should have different boiling points.
The given pair of isomers have different boiling point as they are diastereomers of each other.
(b)
Interpretation:
Whether the compounds in the given pair have the same or different boiling points is to be determined.
Concept introduction:
Isomers are the pair of compounds that have the same formula.
Constitutional isomers are the isomers having the same molecular formula but different connectivity. Constitutional isomers must have different physical and chemical properties.
The configurational isomers are not interconvertible by rotating around a single bond.
Enantiomers are configurational isomers having the same connectivity but cannot be interconvertible by rotation around a single bond. The mirror images of enantiomers are non-superimposable. Since enantiomers have the same connectivity of atoms, they should behave identically.
Diastereomers are configurational isomers that have the same connectivity but are not mirror images of each other. Diastereomers have different physical and chemical properties. Cis-trans isomers are diastereomers of each other.
The boiling point of isomers comes under physical property.
Answer to Problem 5.53P
The given pair of isomers have different boiling points as they are constitutional isomers of each other.
Explanation of Solution
The given pair of compounds is
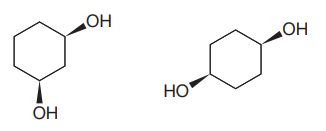
Both the molecules in the given pair have the same molecular formula, so they are isomers.
However, they do not have same connectivity of atoms. In both the compounds, there are two
In the first compound, the
The given pair of isomers have different boiling points as they are constitutional isomers of each other.
(c)
Interpretation:
Whether the compounds in the given pair have the same or different boiling points is to be determined.
Concept introduction:
Isomers are the pair of compounds that have the same formula.
Constitutional isomers are the isomers having the same molecular formula but different connectivity. Constitutional isomers must have different physical and chemical properties.
The configurational isomers are not interconvertible by rotating around a single bond.
Enantiomers are configurational isomers having the same connectivity but cannot be interconvertible by rotation around a single bond. The mirror images of enantiomers are non-superimposable. Since enantiomers have the same connectivity of atoms, they should behave identically.
Diastereomers are configurational isomers that have the same connectivity but are not mirror images of each other. Diastereomers have different physical and chemical properties. Cis-trans isomers are diastereomers of each other.
The boiling point of isomers comes under physical property.
In a Fischer projection, exchanging two groups on an asymmetric carbon atoms gives the opposite stereochemical configuration.
Answer to Problem 5.53P
The given pair of isomers should have different boiling points as they are diastereomers of each other.
Explanation of Solution
The given pair of compounds is
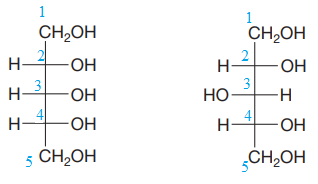
Both the molecules in the given pair have the same molecular formula. They also have the same connectivity. Thus, they are configurational isomers.
Each molecule has three chiral centers at C2, C3, and C4 carbon atoms. Note that in the second compound, the two groups on C3 chiral carbon have been exchanged. This suggests that the stereochemical configurations at C3 carbon atoms in both the compounds are opposite to each other. Remaining stereo centers have the same stereochemical configuration, suggesting that the two compounds are diastereomers of each other. Diastereomers have different physical and chemical properties. Thus, the compounds in the given pair should have different boiling points.
The given pair of isomers have different boiling points as they are diastereomers of each other.
(d)
Interpretation:
Whether the compounds in the given pair have the same or different boiling points is to be determined.
Concept introduction:
Isomers are the pair of compounds that have the same formula.
Constitutional isomers are the isomers having the same molecular formula but different connectivity. Constitutional isomers must have different physical and chemical properties.
The configurational isomers are not interconvertible by rotating around a single bond.
Enantiomers are configurational isomers having the same connectivity but cannot be interconvertible by rotation around a single bond. The mirror images of enantiomers are non-superimposable. Since enantiomers have the same connectivity of atoms, they should behave identically.
Diastereomers are configurational isomers that have the same connectivity but are not mirror images of each other. Diastereomers have different physical and chemical properties. Cis-trans isomers are diastereomers of each other. Cyclic
The boiling point of isomers comes under physical property.
Answer to Problem 5.53P
The given pair of isomers should have different boiling points as they are diastereomers of each other.
Explanation of Solution
The given pair of compounds is
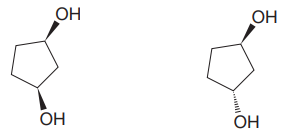
In both the compounds, the
The given pair of isomers have different boiling points as they are diastereomers of each other.
(e)
Interpretation:
Whether the compounds in the given pair have the same or different boiling points is to be determined.
Concept introduction:
Isomers are the pair of compounds that have the same formula.
Constitutional isomers are the isomers having the same molecular formula but different connectivity. Constitutional isomers must have different physical and chemical properties.
The configurational isomers are not interconvertible by rotating around a single bond.
Enantiomers are configurational isomers having the same connectivity but cannot be interconvertible by rotation around a single bond. The mirror images of enantiomers are non-superimposable. Since enantiomers have the same connectivity of atoms, they should behave identically.
The boiling point of isomers comes under physical property.
Answer to Problem 5.53P
The given pair of isomers should have the same boiling point as they are enantiomers of each other.
Explanation of Solution
The given pair of compounds is
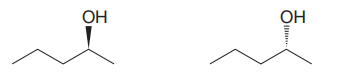
In both the compounds, the
The stereochemical configuration at the chiral center in the first molecule is S as the top-three priority groups are arranged in a counterclockwise manner, and the fourth-priority group is on a dash bond.
The stereochemical configuration at the chiral center in the second molecule is R as the top-three priority groups are arranged in a counterclockwise manner, but the fourth-priority group is on a wedge bond.
Thus, the stereochemical configuration at the chiral centers for two molecules is opposite. This indicates that the two compounds must be enantiomers of each other. Enantiomers have precisely the same physical and chemical properties. Hence the two compounds in the given pair have the same boiling points.
The given pair of isomers should have the same boiling point as they are enantiomers of each other.
Want to see more full solutions like this?
Chapter 5 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- How much more stable is the most stable staggered conformer than the least stable eclipsed conformer?arrow_forwardFatty acids are molecules with a carboxylic acid on one end and long hydrocarbon chain on the other. The sodium salts of fatty acids (such as sodium strearate, shown below) are commonly used in soaps to trap nonpolar dirt and grease particles. Soap does not work well in "hard water, which is water with high concentration of magensium and calcium ions (often found in water obtained from wells) Explain this observation at a molecular level.arrow_forwardWrite the letter T or F inside each box after deciding which statement is true or false. Then on the 3rd box, write the letter of your choice based on the given instructions. A if both statements are true. B if the 1st statement is false and the 2nd statement is true. C if the 1st statement is true and the 2nd statement is false. D if both statements are falsearrow_forward
- Choose the condensed structure for the following line structure. CH3C(CH3)2CH₂O(CH2)4CH3 CH3CH₂C(CH3)2CHO(CH₂)4CH3 CH3CH₂C(CH3)2CH₂O(CH₂)4CH3 CH,CH CH(CH,)CH,O(CH,),CH; CH3CH₂C(CH3)2CH₂OCH(CH2)3CH3arrow_forwardWhich intermolecular force(s) do the following pairs of molecules experience? (Consider asking yourself which molecule in each pair is dominant?) Between acetone and ethanol? Between pentane and hexane? Between pentol and water?arrow_forwardplease answer the 2 questions pleasearrow_forward
- How conformational change involves ring-flipping ?arrow_forwardWhich molecule has the lowest boiling point?arrow_forwardBranching of the hydrocarbon chain lowers the boiling points of the lower homologous series, such as the hydrocarbons and alcohols. Therefore, an assumption can be made that branching lowers the forces attraction in these molecules.arrow_forward
- Make a model of cyclooctane. At least 8 different somewhat stable conformations of this molecule can exist. Draw some of the conformations you can find. Can you find a conformation of this ring that is strain-free? Identify the type of strain present in all possible conformations of this molecule.arrow_forwardTwo isomers of C2H2F2 are shown. Circle the one that you would you expect to have the highest boiling point and explain your reasoning. H. H. H. F isomer A isomer Barrow_forwardPlease answer accuratelyarrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





