
Concept explainers
Draw all possible stereoisomers for each cycloalkane. Label pairs of enantiomers and diastereomers. Label any meso compound.
a. 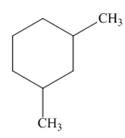 b.
b. 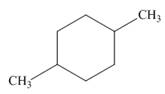 c.
c. 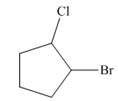
(a)
Interpretation: The possible stereoisomers for the given cycloalkane are to be drawn. The labeling of any enantiomers, diastereomers or meso compound is to be stated.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The stereocenters are carbon atoms on which the interchanging of two atoms or groups results in the formation of new stereoisomers. The stereocenters are also known as stereogenic centers.
Answer to Problem 5.56P
The possible stereoisomers for the given cycloalkane are three. Among the three stereoisomers, the labeling of two enantiomers of the given cycloalkane is
Explanation of Solution
The given cycloalkane is,
Figure 1
Due to the presence of cyclohexane ring attached with two methyl groups, the IUPAC name of this cycloalkane is
Where,
•
Substitute the number of chiral centers of
Thus, the total number of stereoisomers of
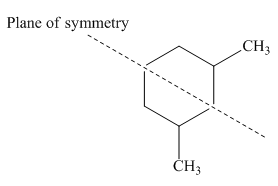
Figure 2
Thus, this compound forms only three stereoisomers instead of four. Among the three stereoisomers of
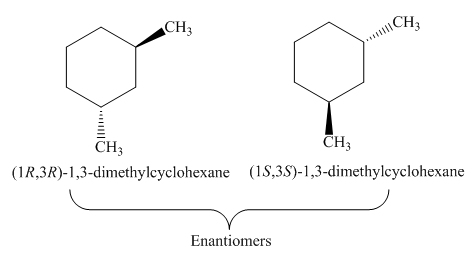
Figure 3
The remaining one stereoisomer of
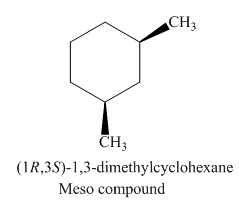
Figure 4
The possible stereoisomers for the given cycloalkane are three. Among the three stereoisomers, the labeling of two enantiomers of the given cycloalkane is
(b)
Interpretation: The possible stereoisomers for the given cycloalkane are to be drawn. The labeling of any enantiomers, diastereomers or meso compound is to be stated.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The stereocenters are carbon atoms on which the interchanging of two atoms or groups results in the formation of new stereoisomers. The stereocenters are also known as stereogenic centers.
Answer to Problem 5.56P
The possible stereoisomers for the given cycloalkane are two. Both are meso compounds and these meso compound are labeled as done as
Explanation of Solution
The given cycloalkane is,
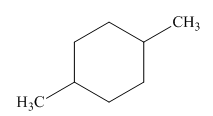
Figure 5
Due to the presence of cyclohexane ring attached with two methyl groups at first and fourth carbon atoms, the IUPAC name of this cycloalkane is
This
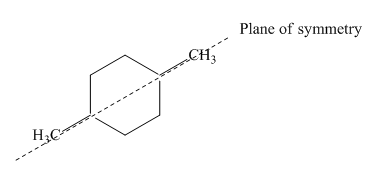
Figure 6
Thus, this compound forms only two stereoisomers one is cis isomer and second is trans isomer. The given compound,
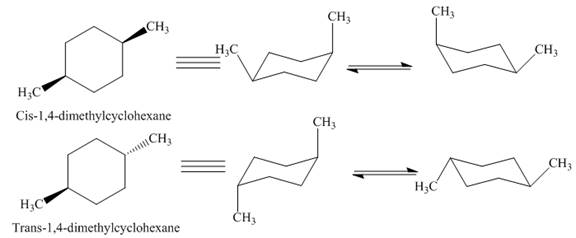
Figure 7
Thus,
The possible stereoisomers for the given cycloalkane are two. Both are meso compounds and these meso compound are labeled as done as
(c)
Interpretation: The possible stereoisomers for the given cycloalkane are to be drawn. The labeling of any enantiomers, diastereomers or meso compound is to be stated.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The stereocenters are carbon atoms on which the interchanging of two atoms or groups results in the formation of new stereoisomers. The stereocenters are also known as stereogenic centers.
Answer to Problem 5.56P
The possible stereoisomers for the given cycloalkane are four. It contains two pairs of enantiomers, the labeling of two pairs of enantiomers of the given cycloalkane is
Explanation of Solution
The given cycloalkane is,
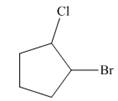
Figure 8
Due to the presence of cyclopentane ring attached with one bromine atom and one chlorine atom at first and second carbon atom respectively, the IUPAC name of this cycloalkane is
Where,
•
Substitute the number of chiral centers of
Thus, the total number of stereoisomers of
The four stereoisomers of
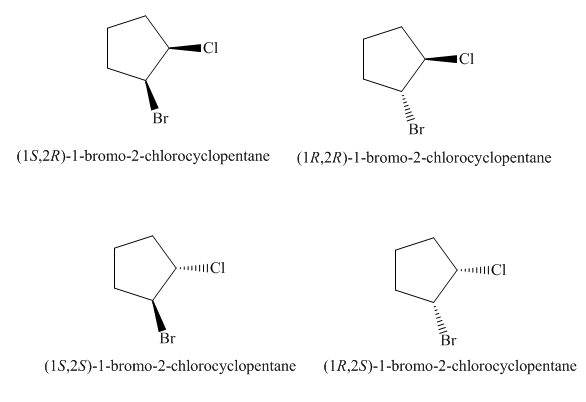
Figure 9
The compound,
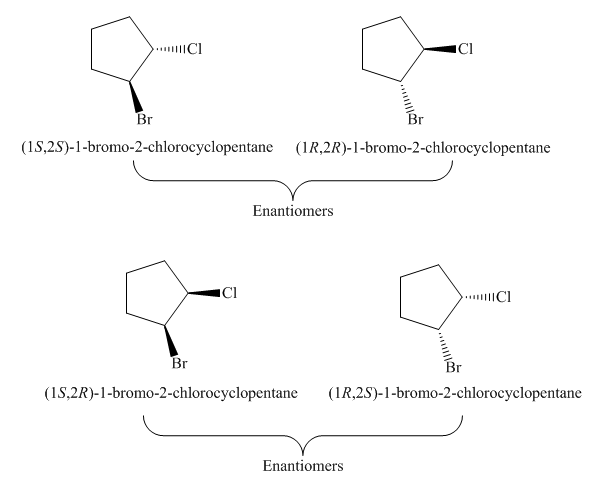
Figure 10
The possible stereoisomers for the given cycloalkane are four. It contains two pairs of enantiomers, the labeling of two pairs of enantiomers of the given cycloalkane is
Want to see more full solutions like this?
Chapter 5 Solutions
Package: Organic Chemistry With Connect 2-semester Access Card
- (a) Draw all stereoisomers formed by monochlorination of the cis and trans isomers of 1,2-dimethylcyclobutane drawn below. (b) How many constitutional isomers are formed in each reaction? (c) Label any pairs of enantiomers formed.arrow_forwardLabel each stereogenic center as R or S (parts d, e and f please)arrow_forwardLabel each pair as constitutional isomers, stereoisomers, or not isomers of each.arrow_forward
- Draw all stereoisomers formed when each alkene is treated with mCPBAarrow_forwardLabel each ring in Figure 7.2 cis or trans.arrow_forwardHow is each compound (B–D) related to A? Choose from enantiomers, diastereomers, identical molecules, constitutional isomers, or not isomers of each other.arrow_forward
- Consider the ball-and-stick model of D, and label E and F as either identical to D or an enantiomer of Darrow_forwardClassify attached compound as identical to A or its enantiomer.arrow_forwardAnswer the following question about compounds A–D (See in attachment) How are the compounds in each pair related? Choose fromconstitutional isomers, stereoisomers, or identical molecules: A and B; A and C; B and D.arrow_forward
- Draw all stereoisomers of Limonene using filled and dashes wedgesarrow_forwardTake a look at the butane conformers below. Identify: (a)Which is an anti conformation in Newman? (b)Which is a Gauche conformation? (c)Which is the more stable Sawhorse conformer? (d)Which has the same potential energy/strain with ALS?arrow_forwardLabel compounds B–D as stereoisomers, conformations, or constitutional isomers of Aarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
