
Concept explainers
An acid-base reaction of
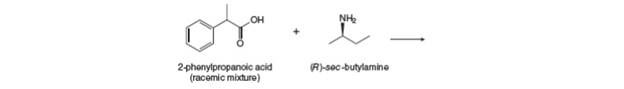
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
ALEKS 360 CHEMISTRY ACCESS
Additional Science Textbook Solutions
Chemistry For Changing Times (14th Edition)
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry: Atoms First
General Chemistry: Atoms First
Chemistry by OpenStax (2015-05-04)
General, Organic, and Biological Chemistry (3rd Edition)
- Following is a retrosynthesis for the coronary vasodilator ganglefene. (a) Propose a synthesis for ganglefene from 4-hydroxybenzoic acid and 3-methyl-3-buten-2-one. (b) Is ganglefene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardIn what way does this steam hydrolysis affect the configuration of the chiral center in pulegone? Assign an R or S configuration to the 3-methylcyclohexanone formed in this reaction.arrow_forwardCompound C undergoes a chemical reaction with a reagent, resulting in the formation of two new compounds. Four potential stereoisomers of the product are shown below. e) Name the type of reaction and provide a possible reagent. f) Clearly assign all the stereocentres found in compound D, E, F and G using Cahn-Ingold-Prelog rules. g) Define the relationship between compound D, E, F and G. h) Identify the two stereoisomers which are likely to be formed from compound C.arrow_forward
- A chiral amine A having the R configuration undergoes Hofmann elimination to form an alkene B as the major product. B is oxidatively cleaved with ozone, followed by CH3SCH3, to form CH2 = O and CH3CH2CH2CHO. What are the structures of A and B?arrow_forwardConsider diastereomers X and Y shown below. Cl diastereomer X Cl HO H OH H Each diastereomer, if treated with sodium ethoxide (NaOCH2CH3), undergoes an E2 reaction. One of the diastereomers undergoes an E2 to make a single compound. The other diastereomers undergoes E2 eliminations leading to two different compounds. Draw the mechanisms for all three of these E2 reactions, being sure to use conformational structures for the substrate in each reaction. Which of the two diastereomers will undergo an E2 reaction at the faster rate? Why? (In organic chemistry it is often said one picture is worth a thousand words. Be sure to include a picture that supports whatever argument you are making.arrow_forwardConsider the E2 elimination of Compound A, and answer the following questions: (a) Label each stereocenter in Compound A with the correct R or S configuration. (b) Draw the structures of the major product with correct stereochemistry. Assume D and H have the same reactivity.arrow_forward
- When pure (S)-lactic acid is esterified by racemic butan-2-ol, the product is 2-butyl lactate, with the following structure:H+CH3 CH COOH CH3 CH CH3 CH CHOH OHCH2CH3 CH2CH3lactic acidC OOH CH O 3butan-2-ol 2-butyl lactate+(a) Draw three-dimensional structures of the two stereoisomers formed, specifying the configuration at each asymmetriccarbon atom. (Using your models may be helpful.)(b) Determine the relationship between the two stereoisomers you have drawnarrow_forwardDescribe how the addition of an isoxazolidinone to a substrate directs the stereochemistry of a reaction.arrow_forwardDraw a line structure clearly showing the stereochemistry of (3S,4S)-4-hydroxy-3,5-dimethylhexanoic acid.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
