
Concept explainers
(a)
Interpretation:
The balanced chemical equation for the process should be written.
Concept Introduction
In order to balance a chemical equation, it is important to define the chemical species in the reactant and product sides. In a balanced chemical equation, for each constituent, atoms on left side of the arrow must be equal to right side.
Interpretation:
The Lewis structure of dinitrogen monoxide should be drawn.
Concept Introduction
Lewis structure is representation of arrangement of valence electrons of atoms in the molecule. The electrons involved in bonding are represented as lines between two atoms and non-bonding electrons are placed on the atom symbol as lone pairs.
(c)
Interpretation:
The mass of N2O should be calculated from 150 g of ammonium nitrate.
Concept Introduction
Mass of a substance can be calculated from number of moles as follows:
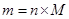
Here, M is molar mass of the substance.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Chemistry For Changing Times (14th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





