
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
6th Edition
ISBN: 9780135765982
Author: Karen Timberlake, William Timberlake
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 5, Problem 82UTC
The chapter sections to review are shown in parentheses at the end of each problem.
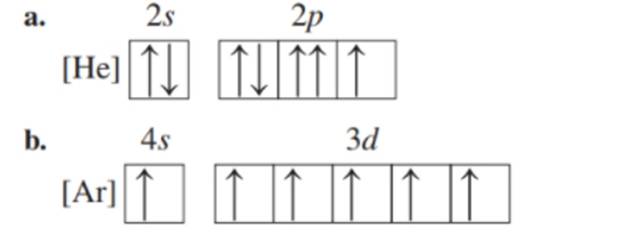
Indicate whether or not each of the following abbreviated orbital diagrams is possible and explain. When possible, indicate the element it represents. (5.4)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Name the element that corresponds to each of the following:(5.4, 5.5, 5.6)a. 1s22s22p63s23p64s13d5b. [Xe]6s24f 145d106p5c. halogen with the highest ionization energy d. Group 2A (2) element with the lowest ionization energye. Period 4 element with the smallest atomic size
Write the abbreviated electron configuration and group numberfor each of the following elements: (5.4)a. Zn b. I c. V d. Sr
28. Completely describe the orbitals found in the fourth
energy level. How many electrons can be found in
a full fourth energy level? Give an example of an
element that has its valence electrons in the fourth
energy
level. (3.3) K/U
Chapter 5 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
Ch. 5.1 - What is meant by the wavelength of UV light?Ch. 5.1 - How are the wavelength and frequency of light...Ch. 5.1 - What is the difference between "white” light and...Ch. 5.1 - Prob. 4PPCh. 5.1 - Ultraviolet radiation (UVB) used to treat...Ch. 5.1 - AM radio waves have a frequency of 8105s1, whereas...Ch. 5.1 - If orange light has a wavelength of 6105cm, what...Ch. 5.1 - A wavelength of 850 nm is used for fiber-optic...Ch. 5.1 - Prob. 9PPCh. 5.1 - Prob. 10PP
Ch. 5.1 - Prob. 11PPCh. 5.1 - Place the following types of electromagnetic...Ch. 5.1 - Prob. 13PPCh. 5.1 - Place the following types of electromagnetic...Ch. 5.2 - What feature of an atomic spectrum indicates that...Ch. 5.2 - How can we explain the distinct lines that appear...Ch. 5.2 - Prob. 17PPCh. 5.2 - Prob. 18PPCh. 5.2 - Prob. 19PPCh. 5.2 - Prob. 20PPCh. 5.3 - Describe the shape of each of the following...Ch. 5.3 - Describe the shape of each of the following...Ch. 5.3 - Match statements 1 to 3 with a to d: 1. They have...Ch. 5.3 - Match statements 1 to 3 with a to d: 1. They have...Ch. 5.3 - Prob. 25PPCh. 5.3 - Indicate the number of each in the following: a....Ch. 5.3 - Prob. 27PPCh. 5.3 - Prob. 28PPCh. 5.4 - Compare the terms electron configuration and...Ch. 5.4 - Compare the terms orbital diagram and electron...Ch. 5.4 - Draw the orbital diagram for each of the...Ch. 5.4 - Draw the orbital diagram for each of the...Ch. 5.4 - Prob. 33PPCh. 5.4 - Write the complete electron configuration for each...Ch. 5.4 - Prob. 35PPCh. 5.4 - Prob. 36PPCh. 5.4 - Prob. 37PPCh. 5.4 - Prob. 38PPCh. 5.4 - Prob. 39PPCh. 5.4 - Give the symbol of the element that meets the...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Use the sublevel blocks on the periodic table to...Ch. 5.5 - Prob. 45PPCh. 5.5 - Use the periodic table to give the symbol of the...Ch. 5.5 - Prob. 47PPCh. 5.5 - Use the periodic table lo give the symbol of the...Ch. 5.5 - Prob. 49PPCh. 5.5 - Prob. 50PPCh. 5.6 - What do the group numbers from IA (1) to 8A (18)...Ch. 5.6 - Prob. 52PPCh. 5.6 - Write the group number using both A/B and 1 to 18...Ch. 5.6 - Write the group number using both A/B and 1 to 18...Ch. 5.6 - Write the valence electron configuration for each...Ch. 5.6 - Prob. 56PPCh. 5.6 - Prob. 57PPCh. 5.6 - Indicate the number of valence electrons in each...Ch. 5.6 - Prob. 59PPCh. 5.6 - Prob. 60PPCh. 5.6 - Prob. 61PPCh. 5.6 - Prob. 62PPCh. 5.6 - Prob. 63PPCh. 5.6 - Select the element in each pair with the higher...Ch. 5.6 - Prob. 65PPCh. 5.6 - Prob. 66PPCh. 5.6 - Prob. 67PPCh. 5.6 - Prob. 68PPCh. 5.6 - Prob. 69PPCh. 5.6 - Prob. 70PPCh. 5.6 - Prob. 71PPCh. 5.6 - Prob. 72PPCh. 5.6 - Which statements completed with a to e will be...Ch. 5.6 - Which statements completed with a to e will be...Ch. 5.6 - Prob. 75PPCh. 5.6 - a. What is the atomic number of Te? b. How many...Ch. 5 - The chapter sections to review are shown in...Ch. 5 - Prob. 78UTCCh. 5 - Prob. 79UTCCh. 5 - Prob. 80UTCCh. 5 - The chapter sections to review are shown in...Ch. 5 - The chapter sections to review are shown in...Ch. 5 - The chapter sections to review are shown in...Ch. 5 - Prob. 84UTCCh. 5 - Prob. 85APPCh. 5 - Prob. 86APPCh. 5 - Prob. 87APPCh. 5 - Prob. 88APPCh. 5 - Prob. 89APPCh. 5 - Prob. 90APPCh. 5 - Prob. 91APPCh. 5 - Prob. 92APPCh. 5 - a. How many 3d electrons are in Fe? (5.4) b. How...Ch. 5 - a. How many 4d electrons are in Cd? (5.4) b. How...Ch. 5 - Write the abbreviated electron configuration and...Ch. 5 - Prob. 96APPCh. 5 - What do the elements Ca, Sr, and Ba have in common...Ch. 5 - Prob. 98APPCh. 5 - Prob. 99APPCh. 5 - Name the element that corresponds to each of the...Ch. 5 - Prob. 101APPCh. 5 - Prob. 102APPCh. 5 - Select the more metallic element in each pair....Ch. 5 - Select the more metallic element in each pair....Ch. 5 - Of the elements Na, P, CI, and F, which (5.6) a....Ch. 5 - Of the elements K, Ca, Br, and Kr, which (5.6) a....Ch. 5 - Prob. 107APPCh. 5 - Prob. 108APPCh. 5 - Prob. 109CPCh. 5 - Prob. 110CPCh. 5 - Prob. 111CPCh. 5 - Prob. 112CPCh. 5 - Prob. 113CPCh. 5 - The following problems are related to the topics...Ch. 5 - The following problems are related to the topics...Ch. 5 - The following problems are related to the topics...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give the symbol of the element that has the (5.6)a. largest atomic size in Period 5b. largest atomic size in Group 2A (2)c. highest ionization energy in Group 8A (18)d. lowest ionization energy in Period 2e. abbreviated electron configuration [Kr]5s24d105p2.arrow_forward4.94 Indicate if each of the following statements is true or false: (4.3) a. The neutron has no net charges. b. Most of the mass of an atom is due to the protons and electrons. c. The total charge of the electrons is equal, but opposite, to the charge of the nucleus. d. The proton and the electron have about the same mass. e. The mass number is the number of protons. 4.10arrow_forward(4.11) Give the values for all four quantum numbers for each electron in the ground state of the nitrogen atom, 7N, writing the positive values for mị and mg first. Present your answer in the exact order as follows: (n1 m mş) for 7N: 1s2 2s² 2p³ subshell (n l m¡ mg) 1s2 2s2 2p3arrow_forward
- Write the group number and draw the Lewis symbol for eachof the following elements: (5.6)a. barium b. fluorine c. krypton d. arsenicarrow_forward(9.3)Match each of the following descriptions with the correct term. Only two electrons with opposing spins are allowed in an orbital. [Choose ] [Choose ] Hund's rule When filling orbitals on the same energy level Pauli's exclusion principle (=degenerate orbitals), electrons fill them singly first (each Aufbau Principle with parallel spin). No two electrons can have the same quantum numbers. [Choose ]arrow_forward4.82 For each of the following, write the symbol and name for X and the number of protons and neutrons. Which are isotopes of each other? (4.4, 4.5) a. 124x A 47 b. 16x 116x 49 C. 50 d. 124x 50 116X e. 48arrow_forward
- 4. 7. 8. 6. 10 11 12 13 14 15 16 17 18 19 20 21 22 23 25 26 A sample of the element Phosphorus is shown to have a mass of 33 and a charge of -3. How many protons would be present in the sample? Phosphorus 15 30.974 A sample of the element Phosphorus is shown to have a mass of 33 and a charge of -3. How would be present in the sample? many electr 24arrow_forward(8.2)Calculate the energy of the green light emitted, per photon, by a mercury lamp with a frequency of 5.49 x 1014 sec¹. O 3.64 x 10-19 J O 1.83 x 10-19 J O 5.46 x 10-19 J O 2.75 x 10-19 J O 4.68 × 10-19 Jarrow_forwardI know I have to somehow convert 275kJ/mol into Joules but I’m not sure how. Could you explain how to solve it?arrow_forward
- Discussion 6 - week 6 Electron Configuration, Periodic Trends (4.7, 4.8), and Nuclear Chemistry (5.1) Part 1: Electron Configurations and Orbital Diagrams of Atoms (4.7, 4.8) 1. Write the abbreviated (short-hand) electron configuration and draw the orbital diagrams for the following ground-state (lowest energy) neutral atoms: A. Sodium (Na) B. Sulfur (S) C. Krypton (Kr) D. Vanadium (V)arrow_forward(10/west 4.4 4.4.1 Calcium reacts with chlorine to form calcium chloride. Draw the Aufbau diagram for a calcium ion. 4.4.2 Write down the chemical symbols of the particles found in the calcium chloride crystal lattice. 5.1 Define the term isotope. Atoms of the same element having the same number of protons, but different number of neutrons. Study the unknown elements A to E below: 5.2 A. 19 9 5.2.1 B. 19 10 C. 20 D. 21 11 E. 19 Which of the elements above are isotopes of each other? 5.2.2 Write down the name of this isotope. 5.3 Calculate the relative atomic mass of copper by using the following isotopes of copper: 636 Copper isotopes: 63 Cu-69% and 65 Cu-31% 163x69)(65x31) Classwork. QUESTION 3 Con 3.1 3.1.1. 3.1.2, 3.1.7 3.1. 100 4347 +2015. 10 100 63,62 5.4 Complete the table below. Write only the answer next to question number 5.4.1-5.4.5. Atomic Number of Number of Number of Element Fluoride ion 5.4.3 Mass number number protons neutrons electrons 5.4.1 9 9 5.4.2 10 25 5.4.4 12…arrow_forward(9.3, 9.4)Identify the element with the ground state electron configuration of [Ar]4s23d¹04p³. O antimony iron tin arsenicarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Quantum Numbers, Atomic Orbitals, and Electron Configurations; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Aoi4j8es4gQ;License: Standard YouTube License, CC-BY
QUANTUM MECHANICAL MODEL/Atomic Structure-21E; Author: H to O Chemistry;https://www.youtube.com/watch?v=mYHNUy5hPQE;License: Standard YouTube License, CC-BY