
Concept explainers
(a)
Interpretation: The elements with the given electronic configuration should be identified as a metal, non-metal or metalloid.
Concept Introduction: The elements which can donate electrons to get the stable electronic configuration are metal, elements that can accept electrons are non-metal. The elements with characteristics of both metals and non-metals are metalloids.
(b)
Interpretation: The element with the smallest atomic size should be identified.
Concept Introduction: According to the periodic trends, the atomic size of the element increases on moving top to bottom in a group due to addition of one shell and decreases on moving left to right in a group due to increase in
(c)
Interpretation: The element with the highest ionization energy should be identified.
Concept Introduction: Ionization energy is defined as the amount of energy required to remove an electron from its outermost shell. According to periodic trends, it decreases on moving top to bottom in a group due to increase in size and it increases on moving left to right in a period due to decrease in size.
(d)
Interpretation: The element with the half-filled sublevel should be identified.
Concept Introduction: The relative energy of orbitals is represented as follows:
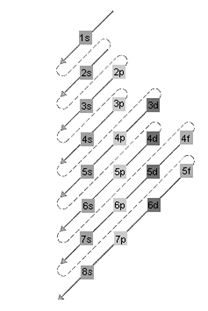
A s orbital can have maximum of 2 electrons, p orbital can have maximum of 6 electrons. Similarly, maximum electrons that a d and f orbital can have are 10 and 14 respectively.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Pearson eText Basic Chemistry -- Instant Access (Pearson+)
- Write the abbreviated electron configuration and group numberfor each of the following elements: (5.4)a. Zn b. I c. V d. Srarrow_forward(9.3)Match each of the following descriptions with the correct term. Only two electrons with opposing spins are allowed in an orbital. [Choose ] [Choose ] Hund's rule When filling orbitals on the same energy level Pauli's exclusion principle (=degenerate orbitals), electrons fill them singly first (each Aufbau Principle with parallel spin). No two electrons can have the same quantum numbers. [Choose ]arrow_forward4. If you were given an antique piece of jewelry and you were told that it was made of solid gold, how could you devise a test to indicate that it was actually made of gold? 6,7.5arrow_forward
- (9.3, 9.4)Identify the element with the ground state electron configuration of [Ar]4s23d¹04p³. O antimony iron tin arsenicarrow_forward4.94 Indicate if each of the following statements is true or false: (4.3) a. The neutron has no net charges. b. Most of the mass of an atom is due to the protons and electrons. c. The total charge of the electrons is equal, but opposite, to the charge of the nucleus. d. The proton and the electron have about the same mass. e. The mass number is the number of protons. 4.10arrow_forward5.96 a. Si b. Se c. Mn d. Sb Write the abbreviated electron configuration and group number for each of the following elements: (5.4) a. Br b. Rh c. Tc d. Raarrow_forward
- (9.6) Which of the following is in the correct order of increasing atomic radius? O Cs > S > Si Cs > Si > S OS > Si > Cs O Si > S > Csarrow_forward(8.2)Calculate the energy of the green light emitted, per photon, by a mercury lamp with a frequency of 5.49 x 1014 sec¹. O 3.64 x 10-19 J O 1.83 x 10-19 J O 5.46 x 10-19 J O 2.75 x 10-19 J O 4.68 × 10-19 Jarrow_forwardWrite the group number and draw the Lewis symbol for eachof the following elements: (5.6)a. barium b. fluorine c. krypton d. arsenicarrow_forward
- 4.121 Complete the following table: (4.3) Namere is ONE O tong of the Element bots he Magnesium Joghe amouse. The Atomic Symbolbo 80 Se 228 F 88 Ra Number of Protons 28 Number of Neutrons 34 14 Number of Electrons Avearrow_forwardDiscussion 6 - week 6 Electron Configuration, Periodic Trends (4.7, 4.8), and Nuclear Chemistry (5.1) Part 1: Electron Configurations and Orbital Diagrams of Atoms (4.7, 4.8) 1. Write the abbreviated (short-hand) electron configuration and draw the orbital diagrams for the following ground-state (lowest energy) neutral atoms: A. Sodium (Na) B. Sulfur (S) C. Krypton (Kr) D. Vanadium (V)arrow_forward6.9.2 What element is represented by the following electronarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





