
Concept explainers
(a)
Interpretation:
The tank with the highest total pressure needs to be determined.
Concept introduction:
According to the
When at two different conditions gases are placed, then to determine the changed variable combined gas law is used. Below is the formula of combined gas law:
Here
- P1 and P2 are the pressure of gases
- V1 and V2 and volume of gases
- n1 and n2 number of moles
- T1 and T2 are the temperature of gases
Moles are known as the ratio of mass and molar mass. Below is the formula:
Here, MM is molar mass and m is the mass.
The kinetic model of gases is accounted for ideal gas behavior. The formula of average translational energy of gas is as below:
Here,
Et = average translational energy of gas
T = temperature in Kelvin
R = Universal gas constant
NA =
Effusion is known as the leakage of gas molecules from high to low pressure region via a pinhole. For any two gas molecules the formula to determine the time needed for effusion is as below:
Here u1 and u2 is the rate of effusion for gas1 and gas 2. MM1 and MM2 is the molar mass for gas1 and gas 2.
Answer to Problem 87QAP
Tank Y
Explanation of Solution
The given tanks are as follows:
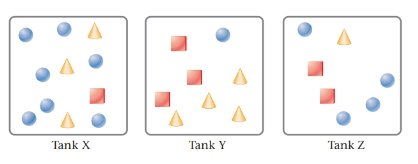
Here, circles represent
The highest total pressure is in the tank Y. Because, tank Y has the maximum amount of gases mixture.
(b)
Interpretation:
The tank containing highest SO2 pressure needs to be determined.
Concept introduction:
According to the ideal
When at two different conditions gases are placed, then to determine the changed variable combined gas law is used. Below is the formula of combined gas law:
Here
- P1 and P2 are the pressure of gases
- V1 and V2 and volume of gases
- n1 and n2 number of moles
- T1 and T2 are the temperature of gases
Moles are known as the ratio of mass and molar mass. Below is the formula:
Here, MM is molar mass and m is the mass.
The kinetic model of gases is accounted for ideal gas behavior. The formula of average translational energy of gas is as below:
Here,
Et = average translational energy of gas
T = temperature in Kelvin
R = Universal gas constant
NA = Avogadro number
Effusion is known as the leakage of gas molecules from high to low pressure region via a pinhole. For any two gas molecules the formula to determine the time needed for effusion is as below:
Here, u1 and u2 is the rate of effusion for gas1 and gas 2. MM1 and MM2 is the molar mass for gas1 and gas 2.
Answer to Problem 87QAP
Tank Y
Explanation of Solution
The given tanks are as follows:
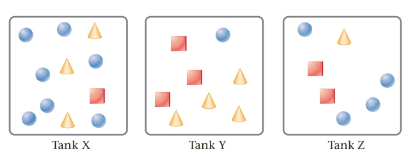
Here, circles represent
Tank Y contains highest partial pressure of SO2 . This is because, Tank Y has the maximum moles of SO2 due to which it will have the maximum partial pressure.
(c)
Interpretation:
The tank with same mass of all the three gases needs to be determined.
Concept introduction:
According to the ideal gas law volume i.e. V, pressure i.e. P, number of moles i.e. m, temperature i.e. t and universal gas constant i.e. R are interrelated as below:
When at two different conditions gases are placed, then to determine the changed variable combined gas law is used. Below is the formula of combined gas law:
Here
- P1 and P2 are the pressure of gases
- V1 and V2 and volume of gases
- n1 and n2 number of moles
- T1 and T2 are the temperature of gases
Moles are known as the ratio of mass and molar mass. Below is the formula:
Here, MM is molar mass and m is the mass.
The kinetic model of gases is accounted for ideal gas behavior. The formula of average translational energy of gas is as below:
Here,
Et = average translational energy of gas
T = temperature in Kelvin
R = Universal gas constant
NA = Avogadro number
Effusion is known as the leakage of gas molecules from high to low pressure region via a pinhole. For any two gas molecules the formula to determine the time needed for effusion is as below:
Here, u1 and u2 is the rate of effusion for gas1 and gas 2. MM1 and MM2 is the molar mass for gas1 and gas 2.
Answer to Problem 87QAP
Tank Z
Explanation of Solution
The given tanks are as follows:
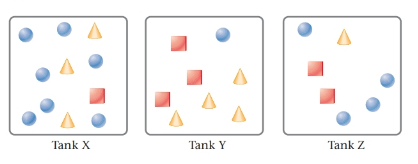
Here, circles represent
Tank Z contain the mass of all three gases same.
(d)
Interpretation:
The tank with the heaviest content needs to be determined.
Concept introduction:
According to the ideal gas law volume i.e. V, pressure i.e. P, number of moles i.e. m, temperature i.e. t and universal gas constant i.e. R are interrelated as below:
When at two different conditions gases are placed, then to determine the changed variable combined gas law is used. Below is the formula of combined gas law:
Here
- P1 and P2 are the pressure of gases
- V1 and V2 and volume of gases
- n1 and n2 number of moles
- T1 and T2 are the temperature of gases
Moles are known as the ratio of mass and molar mass. Below is the formula:
Here, MM is molar mass and m is the mass.
The kinetic model of gases is accounted for ideal gas behavior. The formula of average translational energy of gas is as below:
Here,
Et = average translational energy of gas
T = temperature in Kelvin
R = Universal gas constant
NA = Avogadro number
Effusion is known as the leakage of gas molecules from high to low pressure region via a pinhole. For any two gas molecules the formula to determine the time needed for effusion is as below:
Here u1 and u2 is the rate of effusion for gas1 and gas 2. MM1 and MM2 is the molar mass for gas1 and gas 2.
Answer to Problem 87QAP
Tank Y
Explanation of Solution
The given tanks are as follows:
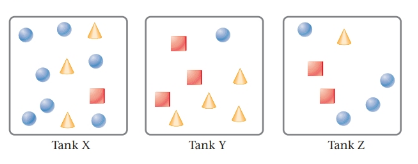
Here, circles represent
Tank Y contains the heaviest content. This is due to the number of moles of SO2 is highest in tank Y.
Want to see more full solutions like this?
Chapter 5 Solutions
Chemistry: Principles and Reactions
- How does hydraulic fracturing differ from previously used techniques for the recovery of natural gas from the earth?arrow_forwardYou have two pressure-proof steel cylinders of equal volume, one containing 1.0 kg of CO and the other containing 1.0 kg of acetylene, C2H2. (a) In which cylinder is the pressure greater at 25 C? (b) Which cylinder contains the greater number of molecules?arrow_forwardWhen hydrogen peroxide decomposes, oxygen is produced: 2H2O2(aq)2H2O+O2(g)What volume of oxygen gas at 25C and 1.00 atm is produced from the decomposition of 25.00 mL of a 30.0% (by mass) solution of hydrogen peroxide (d=1.05g/mL)?arrow_forward
- What volume (in liters) of O2, measured at standard temperature and pressure, is required to oxidize 0.400 mol of phosphorus (P4)? P4(s) + 5 O2(g) P4O10(s)arrow_forwardWhat possible uses exist for the natural gas liquids that are removed from natural gas during its processing?arrow_forward95 Some engineering designs call for the use of compressed air for underground work. If water containing iron(II) ions is present, oxygen in the compressed air may react according to the following unbalanced net ionic equation: Fe2++H++O2Fe3++H2O (a) Write the balanced net ionic equation. Remember that the amounts of each substance and the charges must balance. (b) Assume all of the oxygen from 650 L of compressed air at 15°C and 6.5 atm is lost by this reaction. What mass of water would be produced? (The mole fraction of oxygen in air is about 0.21.) (c) What will be the final pressure after the loss of the oxygen?arrow_forward
- Consider the following sketch. Each square in bulb A represents a mole of atoms X. Each circle in bulb B represents a mole of atoms Y. The bulbs have the same volume, and the temperature is kept constant. When the valve is opened, atoms of X react with atoms of Y according to the following equation: 2X(g)+Y(g)X2Y(g)The gaseous product is represented as and each represents one mole of product. (a) IfP A=2.0 atm, what is P8 before the valve is opened and the reaction is allowed to occur? What is P A+P B? (b) Redraw the sketch to represent what happens after the valve is opened. (c) What is PA? What is PB? What is P A+P B? Compare your answer with the answer in part (a).arrow_forwardYou have a gas, one of the three known phosphorus-fluorine compounds (PF3, PF3, and P2F4). To find out which, you have decided to measure its molar mass. (a) First, yon determine that the density of the gas is 5.60 g/L at a pressure of 0.971 atm and a temperature of 18.2 C. Calculate the molar mass and identify the compound. (b) To check the results from part (a), you decide to measure the molar mass based on the relative rales of effusion of the unknown gas and CO2. You find that CO2 effuses at a rate of 0.050 mol/min, whereas the unknown phosphorus fluoride effuses at a rate of 0.028 mol/min. Calculate the molar mass of the unknown gas based on these results.arrow_forwardGiven that a sample of air is made up of nitrogen, oxygen, and argon in the mole fractions 0.78 N2, 0.21 O2, and 0.010 Ar, what is the density of air at standard temperature and pressure?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





