
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5.4, Problem 5.23P
Propose a mechanism for each of the following reactions:
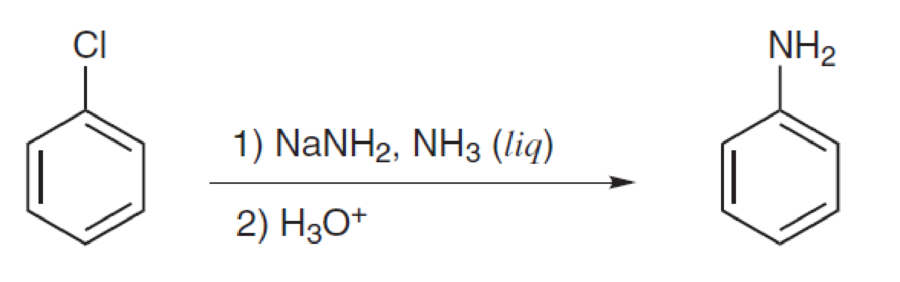
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose the mechanisms for each step of the synthesis below.
propose mechanisms for the following reactions.
Propose a possible reaction mechanism for the following transformation. Draw out the steps with the correct reaction arrows.
Chapter 5 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 5.1 - Prob. 5.2PCh. 5.1 - Prob. 5.3PCh. 5.1 - Prob. 5.4PCh. 5.1 - Prob. 5.5PCh. 5.1 - Prob. 5.6PCh. 5.1 - Prob. 5.7PCh. 5.2 - Propose a mechanism for each of the following...Ch. 5.2 - Propose a mechanism for each of the following...Ch. 5.2 - Propose a mechanism for each of the following...Ch. 5.3 - Prob. 5.13P
Ch. 5.3 - Prob. 5.14PCh. 5.3 - Prob. 5.15PCh. 5.3 - Prob. 5.16PCh. 5.3 - Prob. 5.17PCh. 5.3 - Prob. 5.18PCh. 5.4 - Propose a mechanism for each of the following...Ch. 5.4 - Propose a mechanism for each of the following...Ch. 5.4 - Propose a mechanism for each of the following...Ch. 5.4 - Propose a mechanism for each of the following...
Additional Science Textbook Solutions
Find more solutions based on key concepts
A compound that contains only C and H was burned in excess O2 to give CO2 and H2O. When 0.270 g of the compound...
General Chemistry: Atoms First
PRACTICE 1.3 The melting point of table salt is 1474oF. What temperature is this on the Celsius and Kelvin scal...
CHEMISTRY-TEXT
39. What are the units of k for each type of reaction?
a. first-order reaction
b. second-order reaction
c...
Chemistry: Structure and Properties
The hormone estrogen is produced in the ovaries of females and elsewhere in the body in men and postmenopausal ...
Elementary Principles of Chemical Processes, Binder Ready Version
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The Meerwein-Ponndorf-Verley reaction involves reduction of a ketone by treatment with an excess of aluminum triisopropoxide, [(CH3)2CHO]3Al. The mechanism of the process is closely related to the Cannizzaro reaction in that a hydride ion acts as a leaving group. Propose a mechanism.arrow_forwardShow the products you would obtain by acid-catalyzed reaction of cyclohexanone with ethylamine, CH3CH2NH2, and with diethylamine, (CH3CH2) 2NH.arrow_forwardIf methanol rather than water is added at the end of a Hell-Volhard-Zelinskii reaction, an ester rather than an acid is produced. Show how you would carry out the following transformation, and propose a mechanism for the ester-forming step.arrow_forward
- Treatment of p-tert-butylphenol with a strong acid such as H2SO4 yields phenol and 2-methylpropene. Propose a mechanism.arrow_forwardTreatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forwardPropose a structure for the product with formula C9H17N that results when 2-(2-cyanoethyl)cyclohexanone is reduced catalytically.arrow_forward
- Knoevenagel condensation is a reaction involving an active methylene compound (a CH2 flanked by two electron-withdrawing groups) and an aldehyde and ketone. Propose a mechanism for the reaction below.arrow_forwardSolvolysis of the following bicyclic compound in acetic acid gives a mixture of products, two of which are shown. The leaving group is the anion of a sulfonic acid, ArSO3H. A sulfonic acid is a strong acid, and its anion, ArSO3, is a weak base and a good leaving group. Propose a mechanism for this reaction.arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forward
- Following are the steps in one of the several published syntheses of frontalin, a pheromone of the western pine beetle. (a) Propose reagents for Steps 18. (b) Propose a mechanism for the cyclization of the ketodiol from Step 8 to frontalin.arrow_forwardPropose a mechanism for the following reactionsarrow_forwardpropose a mechanism for the following transformationsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License