
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 5.3, Problem 5.18P
Interpretation Introduction
Interpretation:
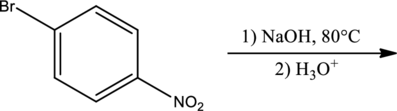
Products for the given reaction has to be predicted.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write down the mechanism of the reactions given below and find the resulting product.
Propose a mechanism for the observed product based on what you know about SN1 and SN2-type mechanisms.
Propose a mechanism and predict the products for each reaction. For reactions where more then one product can form, draw all possible products and predict which would be the major product.
Chapter 5 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 5.1 - Prob. 5.2PCh. 5.1 - Prob. 5.3PCh. 5.1 - Prob. 5.4PCh. 5.1 - Prob. 5.5PCh. 5.1 - Prob. 5.6PCh. 5.1 - Prob. 5.7PCh. 5.2 - Propose a mechanism for each of the following...Ch. 5.2 - Propose a mechanism for each of the following...Ch. 5.2 - Propose a mechanism for each of the following...Ch. 5.3 - Prob. 5.13P
Ch. 5.3 - Prob. 5.14PCh. 5.3 - Prob. 5.15PCh. 5.3 - Prob. 5.16PCh. 5.3 - Prob. 5.17PCh. 5.3 - Prob. 5.18PCh. 5.4 - Propose a mechanism for each of the following...Ch. 5.4 - Propose a mechanism for each of the following...Ch. 5.4 - Propose a mechanism for each of the following...Ch. 5.4 - Propose a mechanism for each of the following...
Knowledge Booster
Similar questions
- Give a detailed reaction mechanism for the reaction expected to occur when 2-bromo-2-methylpentane is heated with sodium methoxide. Draw clear structural formulas of all relevant species and use curved arrows to represent electron flow. Also indicate which step is likely to be rate-determining. The answer you sent before will be used for this question.arrow_forwardWhat will be the resulting organic products’ structure when the following reactions occur and determine the mechanism (SN1, SN2, E1 or E2)? 1-brochloro-1methylcyclohexane with Potassium Hydroxide (encircle the major product among the two products)arrow_forwardThe following reaction is slow and gives an unusual product. Propose a mechanism for the observed product based on what you know about SN1 and SN2-type mechanisms.arrow_forward
- Decide which compounds from the list below are best suited for nucleophilic addition reactions and which ones are more appropriate for nucleophilic substitution reactions.arrow_forwardThis question refers to the following reaction: (SEE IMAGE BELOW) i) Determine whether the main reaction to form F is a substitution or an elimination reaction. Explain your choice in no more than two sentences. ii) Draw the structure of the product F. iii) What is the mechanism of the above reaction (i.e. SN1, SN2, E1 or E2)? Explain your choice in one sentence based on your answer in (i) above. iv) Using curly arrows show the mechanism for the reaction to give F.arrow_forwardThe reaction of 2,2-dimethyl-1-propanol with HBr is very slow and forms 2-bromo-2-methylbutane as the main product (see picture) Come up with a mechanism to explain these observationsarrow_forward
- What is the mechanism for the entire reaction below?arrow_forwardExplain the possible mechanism (Sn1/Sn2/E1/E2) given the reaction. Show the transfer of electrons/groups, and name the product.arrow_forwardBelow is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?arrow_forward
- For each reaction, give the expected substitution product and predict whether the mechanism will be first order (SN1) or second order (SN2): a) 2-chloro-2-methylbutane + CH3COOH b) isobutyl bromide + NaOMe c) 1-iodo-1-methylcyclohexane + CH3CH2OHarrow_forwardFor each of the substrates below, identify whether: (A) the rate of substitution doesn't depend on nucleophile concentration and (B) the product distribution from substitution gives a 50/50 mix of enantiomers. If you answered "yes" for the first substrate, draw the intermediate that forms during a nucleophilic substitution reaction in the space below the tablearrow_forwardFunctional groups such as alkynes react the same in complex molecules as they do in simpler structures. The following example of alkyne reaction were taken from syntheses carried out in the research group of E. J. Corey at Harvard University. You can assume that the reactions listed involve only the alkyne, not any of the functional groups present in the molecules. Draw the expected products for the following reaction .arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
