
Concept explainers
Interpretation:
The suitable compound used to produce the given products after reaction with ozone and dimethyl sulfide should be identified.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
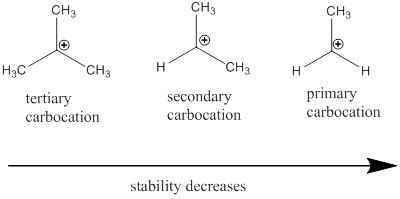
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
Ozonolysis Reaction: It is an oxidative reaction which is used to oxidize the carbon-carbon double and triple bond.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ORGANIC CHEMISTRY PKG *FULL YEAR* >CI<
- Heck reactions take place with alkynes as well as alkenes. The following conversion involves an intramolecular Heck reaction followed by an intermolecular Heck. Propose structural formulas for the palladium-containing intermediates involved in this reaction.arrow_forwardWittig reactions with the following -chloroethers can be used for the synthesis of aldehydes and ketones. (a) Draw the structure of the triphenylphosphonium salt and Wittig reagent formed from each chloroether. (b) Draw the structural formula of the product formed by treating each Wittig reagent with cyclopentanone. Note that the functional group is an enol ether or, alternatively, a vinyl ether. (c) Draw the structural formula of the product formed on acid-catalyzed hydrolysis of each enol ether from part (b).arrow_forwardHow might you replace a halogen substituent by a deuterium atom if you wanted to prepare a deuterated compound?arrow_forward
- Birch Reduction of toluene leads to a product, X with the molecular formula C7H10. After ozonolysis of X, the two compounds, 3-oxobutanal and malonaldehyde are formed. what is the structure of xarrow_forwardWhat is the major product between the reaction of 2-methoxypentane and HBr reagent?arrow_forwardWhat is the major product formed from reacting cyclohexene in aqueous Br2, followed by treatment with potassium tert-butoxide?arrow_forward
- What is the major organic product obtained from the following reaction? 1, 2, 3, or 4?arrow_forwardA hydrocarbon, C6H12, on ozonolysis gives only one product which does not give silver mirror with Tollens' reagent. The hydrocarbon is ?arrow_forwardWhat products are obtained from the reaction of cyclohexene oxide with a. methoxide ion? b. methylamine?arrow_forward
- what are the reagents used to covert butane into (e)-2-butene and then into C4H8O? How would C4H8O be structured?arrow_forwardGive the balanced equation of acetylene and the following reactions • Combustion • dilute KMnO4 • Ammoniacal CuCl / Ammonical AgNO3arrow_forwardHow will you bring about the following conversions? (i)Aniline to chlorobenzene (ii) 2-Methyl-1-propene to 2-chloro-2-methylpropane (iii) Ethyl chloride to propanoic acid (iv) But-1-ene to n-butyliodide (v) Benzene to Phenyl chloromethane. (vi) Tert-butylbromide to isobutylbromide.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

