
Concept explainers
All molecules undergo vibrational motions.
Where n is a quantum number
Interpretation:
The first three vibrational energy levels for
l is to be drawn. The energy required for the transition of molecule from ground state to first excited state is to be determined, and “the reason for lowest vibrational energy in ground state is not zero, but it is equivalent to
” is to be justified by using Heisenberg Principle.
Concept introduction:
The energy of a photon can be expressed as follows:
Here, E is the energy of photon,
Heisenberg uncertainty principle explains that the product of uncertainty in position and momentum of particle cannot be less than
Here,
denotes uncertainty in position,
denotes uncertainty in momentum, and
denotes Planck’s constant
Answer to Problem 112AP
Solution:
a)
(b)
(c) Consider the diatomic molecule
This is disallowed by the Heisenberg uncertainty principle.
Explanation of Solution
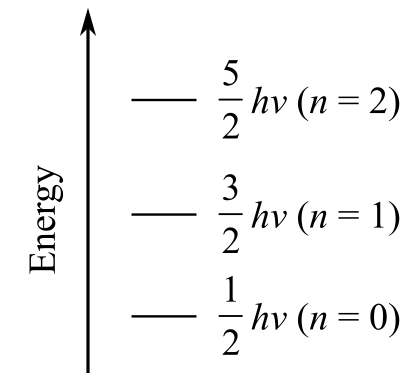
a) Plot the first three vibrational energy levels for HCl
The vibrational energy of diatomic molecules is given as
The first three vibrational energy levels n are
For
For
For
The vibrational energy for the first three energy levels is

b) The vibrational energy required to excite HCl molecule from ground state to first exited state.
The vibrational energy for the ground state and first excited state are as follows:
For the transition from ground state
Now, substitute the value
and
in the above equation
c) Justify the prediction “that the lowest vibrational energy in the ground state is not zero, but it is equivalent to
Consider the diatomic molecule
As the two atoms are bonded to each other, the uncertainty in position, that is,
Thus,
So, this is disallowed by the Heisenberg uncertainty principle.
Want to see more full solutions like this?
Chapter 6 Solutions
Aleks 360 Access Card 1 Semester For Introductory Chemistry
Additional Science Textbook Solutions
Fundamentals of Heat and Mass Transfer
Basic Chemistry (5th Edition)
Chemistry: The Central Science (14th Edition)
CHEMISTRY-TEXT
Principles of General, Organic, Biological Chemistry
- Consider burning ethane gas, C2H6 in oxygen (combustion) forming CO2 and water. (a) How much energy (in J) is produced in the combustion of one molecule of ethane? (b) What is the energy of a photon of ultraviolet light with a wavelength of 12.6 nm? (c) Compare your answers for (a) and (b).arrow_forwardSuppose that the microwave radiation has a wavelength of 12.4 cm. How many photons are required to heat 285 mL of coffee from 25.0 ∘C∘C to 62.0 ∘C∘C? Assume that the coffee has the same density, 0.997 g mL−1, and specific heat capacity, 4.184 J g−1 K−1, as water over this temperature range. Express the number of photons numerically.arrow_forwardPhotosynthesis uses 660nm light to convert CO2 and H2O into C6H12O6 and H2O Calculate the frequencyarrow_forward
- What is the frequency of the photons emitted by hydrogen atoms when they undergo transitions from n = 5 to n = 3?arrow_forwardCa lcu late the size of the quantum involved in the excitation of (a) an electronic motion of frequency 1.0 x 1015 Hz. (b) a molecular vibration of period 20 Is. (c) a pendulum of period 0.50 s. Express the results in joules and in kilojoules per mole.arrow_forward2. Calculate the wavelenth , in meters, of microwave radiation that corresponds to an energy of 2.63 J/mol of photons.arrow_forward
- a. What information does the term symbol 3F4 provide about the angular momentum of an atom? b. What information does the term symbol 1D2 provide about the angular momentum of an atom?arrow_forwardUltraviolet light with a wavelength of 400 nm falls on a specific metal surface. The maximum value of the radiated kinetic energy of photoelectrons is 1.10 eV. What is the maximum kinetic energy Kmax of photoelectrons when the 350 nm wavelength light falls on the same surface?arrow_forwardWhat is the energy of a single photon of microwave radiation with a wavelength of 0.0618 meters? Your answer should have 3 sig figs.arrow_forward
- Calculate the frequiency of the light emitted by hydrogen atoms when they undergo transitions from n=5 to n=3? In what region of the electromagnetic spectrum does the radiation occur?arrow_forwardNeutrons, like electrons and photons, are particle-waves whose diffraction patterns can be used to determine the structures of molecules. Calculate the kinetic energy (in J) of a neutron with wavelength 51.1 pm.arrow_forwardCalculate the energy of the quantum involved in the excitation of (i) an electronic oscillation of period 1.0 fs, (ii) a molecular vibration of period 10 fs, (iii) a pendulum of period 1.0 s. Express the results in joules and kilojoules per mole.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning


