
CAMPBELL BIOLOGY:CONCEPTS..W/JUNGLE>IC
15th Edition
ISBN: 9781269930741
Author: Reece
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 13TYK
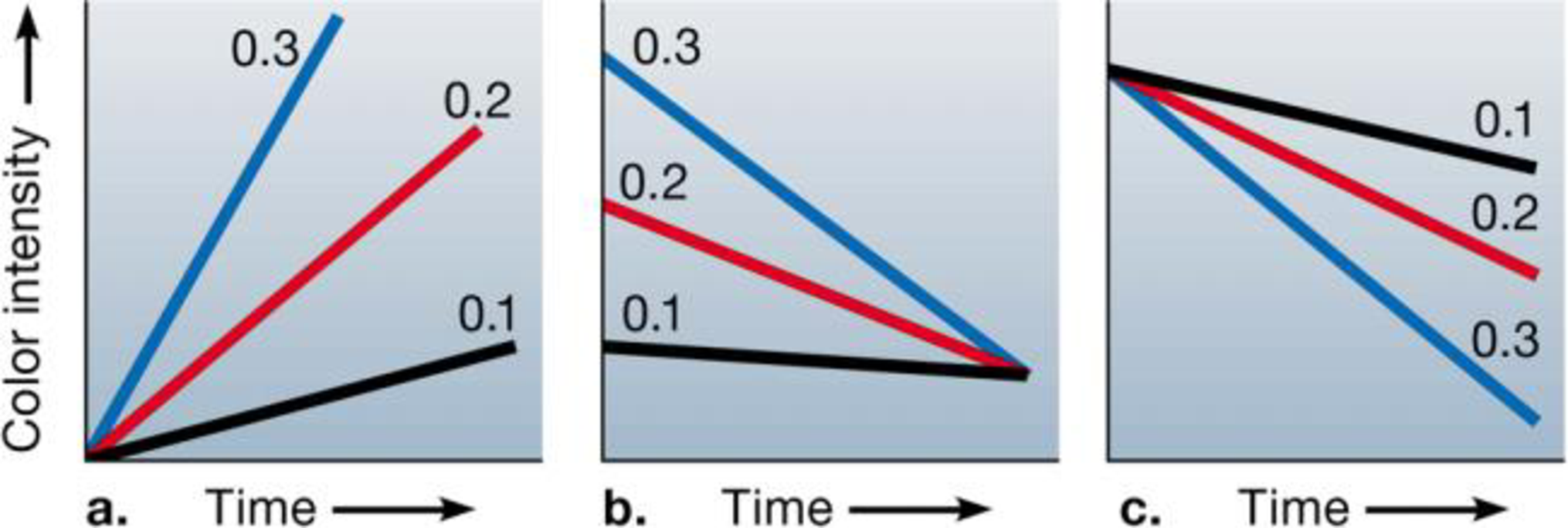
In the citric acid cycle, an enzyme oxidizes malate to oxaloacetate, with the production of NADH and the release of H+. You are studying this reaction using a suspension of bean cell mitochondria and a blue dye that loses its color as it takes up H+. You set up reaction mixtures with mitochondria, dye, and three different concentrations of malate (0.1 mg/L, 0.2 mg/L, and 0.3 mg/L). Which of the following graphs represents the results you would expect, and why?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Interested in studying ethanol fermentation, you prepare two solutions of yeast extract that contains the cytosolic contents of Saccharomyces cerevisiae. You add glucose to both extracts at time zero, but to one (in red) you also add inorganic phosphate (added in two batches). Using a gas sensor, you monitor the CO2 generated by the two solutions over time and obtain the following data:
The citric acid cycle generates NADH and FADH2, which are then used in the process of oxidative phosphorylation to make ATP. If the citric acid cycle (which does not use oxygen) and oxidative phosphorylation are separate processes, as they are, then why is it that the citric acid cycle stops almost immediately when O2 is removed?
Interested in studying ethanol fermentation, you prepare two solutions of yeast extract that contains the cytosolic contents of Saccharomyces cerevisiae. You add glucose to both extracts at time zero, but to one (in red) you also add inorganic phosphate (added in two batches). Using a gas sensor, you monitor the CO2 generated by the two solutions over time and obtain the following data:
a. Why is more CO2 generated upon the addition of phosphate?
b. You become worried that the flasks would burst open to the build up in gas pressure. What other molecules could you add to the extract to slow down CO2 generation even in the presence of glucose and Pi?
Chapter 6 Solutions
CAMPBELL BIOLOGY:CONCEPTS..W/JUNGLE>IC
Ch. 6 - Fill in the blanks in this summary map to help you...Ch. 6 - A biochemist wanted to study how various...Ch. 6 - In glycolysis, _____ is oxidized and _____ is...Ch. 6 - Which of the following is the most immediate...Ch. 6 - Which of the following is a true distinction...Ch. 6 - The poison cyanide binds to an electron carrier...Ch. 6 - In which of the following is the first molecule...Ch. 6 - Which of the three stages of cellular respiration...Ch. 6 - Compare and contrast fermentation as it occurs in...Ch. 6 - Prob. 10TYK
Ch. 6 - Prob. 11TYKCh. 6 - Prob. 12TYKCh. 6 - In the citric acid cycle, an enzyme oxidizes...Ch. 6 - ATP synthase enzymes are found in the prokaryotic...Ch. 6 - 15. SCIENTIFIC THINKING Several studies have found...Ch. 6 - For a short time in the 1930s, some physicians...Ch. 6 - Explain how the mechanism of brown fat metabolism...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Which of the following statements is NOT true about the electron transport chain (ETC) and oxidative phosphorylation? A. Oxidative phosphorylation requires the enzyme complexes to be soluble in the mitochondrial matrix. B. If the proton gradient is too high, electrons will not move through the ETC. C. The movement of electrons down the ETC only happens if protons are pumped out of the mitochondrial matrix. D. Oxidative phosphorylation requires the mitochondrial intermembrane space to be more positively charged than the matrix. E. The free energy of the proton gradient can be used to create high energy bonds.arrow_forwarda. Starting with acetyl-CoA in the mitochondria, how many ATP molecules are needed to synthesize a 14 C fatty acid? A. 3 B. 6 C. 9 D. 12 b. How many FADH2 molecules will be obtained from the complete oxidation of Myristic acid to CO2 ? A. 13 B. 11 C. 15 D. 17 c. If the complete oxidation of a saturated fatty acid result in the production of 82 ATP molecules, how many ATP molecules will be obtained from a mono-unsaturated fatty acid of similar length? A. 80.5 B. 82 C. 84.5 D. 83.5arrow_forwardAt the very end of cellular respiration (glycolysis followed by pyruvate oxidation, the citric acid cycle, and electron transport/ATP synthesis), a. In what molecules are all of the carbons from the glucose? b. In what molecules are many of the electrons from the glucose? c. In what molecules is much of the free energy that was harvested from the glucose?arrow_forward
- Which of the following is the second step of Citric Acid Cycle? Select one: a. Isocitrate and then decarboxylated and oxidized to produce alpha-ketoglutarate, Carbon dioxide and NADH b. Succinyl-CoA becomes Succinate and forms one ATP molecule and Coenzyme A-SH c. alpha-ketoglutarate is oxidized and decarboxylated to produce Succinyl-CoA, Carbon dioxide and NADH d. Malate is oxidized to become oxaloacetate forming NADH e. Fumarate is combined with water to become Malate f. Citrate is rearranged to become Isocitrate g. Pyruvate is decarboxylated to become acetyl-CoA producing NADH and Carbon dioxide h. Oxaloacetate combines with the acetyl from acetyl-CoA to produce Citric acid(citrate) i. Succinate is oxidized to become fumarate forming FADH2arrow_forwardWhich of the following drives the electron transfer from coenzyme Q to cytochrome c, which also results to the pumping of protons from the mitochondrial matrix to the intermembrane space? Complex I Complex II Complex III Complex IV If 2 molecules of FADH2 are oxidized during oxidative phosphorylation, how many molecules of ATP are synthesized? Your answer should be a numerical input.arrow_forwardIn biochemistry, the exergonic process ofconverting glucose and oxygen to carbon dioxide and water in aerobic metabolism can be considered the reverse of photosynthesis,in which carbon dioxide and water are converted to glucose andoxygen. Do you expect both processes to be exergonic, both endergonic, or one exergonic and one endergonic? Why? Would youexpect both processes to take place in the same way? Why?arrow_forward
- Which of the following statements about cellular respiration is TRUE in eukaryotes? A. For every molecule of NADH oxidized in the electron transport chain, 1 molecule of ATP is produced. B. ATP synthase harnesses the flow of protons (hydrogen ions = H+) from the mitochondrial matrix to the intermembrane space to produce ATP. C. ATP synthase transduces the flow of protons (hydrogen ions = H+) from the intermembrane space to the mitochondrial matrix into kinetic (mechanical) energy D. H2O is the final electron acceptor in the electron transport chain, being oxidized to O2 and H+. E. All of the above are truearrow_forwardTo reduce six molecules of carbon dioxide to one molecule of glucose via photosynthesis, how many molecules of NADPH and ATP are required?arrow_forwardin one of the reactions in the electron transport chain, complex I transfer electrons to coenzyme Q. which molecule is reduced and which is oxidized in this reaction? a. complex I is reduced b/c it loses electrons, and Q is oxidized b/c it gains electrons b. complex I is oxidized b/c it loses electrons, and Q is reduced b/c it gains electrons c. complex I is reduced b/c it gains electrons, and Q is oxidized b/c it loses electrons d. complex 1 and Q are both oxidized b/c elections are moved from one molecule to the otherarrow_forward
- The Krebs cycle occurs in the Select one: a. cytoplasm, as the compound acetyl CoA enters. b. mitochondria, as the compound pyruvate (C3) enters. c. mitochondria, as the compound acetyl CoA enters. d. cytoplasm, as the compound pyruvate (C3) enters.arrow_forwardWhich of the following statements concerning the complete oxidation of FADH2 in the electron transport chain is NOT true? a. In the final step, electrons from cytochrome c to O2 reducing it to H2O in complex IV, and four protons are transported from the intermembrane space to the matrix. b. In the first step, electrons from FADH2 are transferred in complex II to ubiquinone, which does not transport any proton across the inner mitochondrion membrane. c. In the second step, complex III transfers the electrons from ubiquinone to cytochrome c, and four protons are transported from the matrix to the intermembrane space. d. The complete oxidation of FADH2 causes transfer of 6 protons and yields two ATP.arrow_forwardWhen you stop running the citric acid cycle and have sufficient ATP, citrate can build up in the mitochondria, what happens to this excess citrate? What are the two primary intermediate molecules produced by citrate in the cytoplasm? What storage molecule is made?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Intro to Food Microbiology; Author: A professor pressing record;https://www.youtube.com/watch?v=vg8fSmk0dVU;License: Standard youtube license