
Chemistry - Modified MasteringChemistry
7th Edition
ISBN: 9780133892321
Author: McMurry
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 6.10A
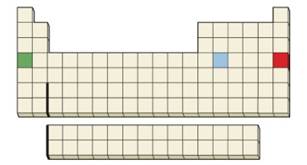
Conceptual APPLY 6.10 Which of the indicated three elements has the least favorable Eea, and which has the most favorable Eea?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
6. (8) In general, as effective charges increases, ionization energy (/,) also increases. Explain why it requires much higher energy (AH- 1405 kJ/Mol) for element Nitrogen (N) than element
oxygen (O) (AH= 1314 kJ/Mol)? (Hint: You may need to consider writing their orbital diagram before proceeding to answer this question).
For each of the following pairs of elements (C and N) and (Ar and Br), pick theatom in the pair that has
C or N Ar or Br(i) the more exothermic electron affinity:(ii) the higher ionization energy:(iii) the larger atomic radius:
Circle the approximate part or parts of the periodic table where the following elements appear.
(a) Elements with the smallest values of Ea
(b)
Elements with the largest atomic radil
(c)
Elements with the most negative values of Ea
Drag the appropriate labels to their respective targets. Labels with E should be placed inside the table.
Group 2
Group 2
Group 1
Group T
the
smallest
values of
Ea
Group 1
Group t
the largest
atomic
radi
Group-1
Group 1
the most
negative
values of
E
Group 2
Group
Group 1
Group T
Group-1
Group T
Group 1
Group 1
Group t
Group T
Group 1
Group 1
Group T
Group 1
Reset Help
Group 2
Chapter 6 Solutions
Chemistry - Modified MasteringChemistry
Ch. 6 - Prob. 6.1PCh. 6 - APPLY 6.2 Which of the following sets of ions are...Ch. 6 - Prob. 6.3PCh. 6 - Conceptual APPLY 6.4 Which of the following...Ch. 6 - Prob. 6.5PCh. 6 - Prob. 6.6ACh. 6 - Prob. 6.7PCh. 6 - Conceptual APPLY 6.8 The figure on the right...Ch. 6 - Prob. 6.9PCh. 6 - Conceptual APPLY 6.10 Which of the indicated three...
Ch. 6 - Prob. 6.11PCh. 6 - Prob. 6.12ACh. 6 - Prob. 6.13PCh. 6 - APPLY 6.14 Calculate the energy of electrostatic...Ch. 6 - Prob. 6.15PCh. 6 - Conceptual APPLY 6.16 One of the following...Ch. 6 - Prob. 6.17PCh. 6 - PROBLEM 6.18 Compare the following two ionic...Ch. 6 - PROBLEM 6.19 An ionic liquid consisting of a bulky...Ch. 6 - Where on the periodic table would you find the...Ch. 6 - Which of the following spheres is likely to...Ch. 6 - Circle the approximate part or parts of the...Ch. 6 - Prob. 6.23CPCh. 6 - This figure represents the successive ionization...Ch. 6 - In the following drawings, red spheres represent...Ch. 6 - Which of the following drawings is more likely to...Ch. 6 - Prob. 6.27CPCh. 6 - Which of the following alkali metal halides has...Ch. 6 - Which of the following alkali metal halides has...Ch. 6 - Three binary compounds are represented on the...Ch. 6 - Given the following values for the formation of...Ch. 6 - What is the difference between a covalent bond and...Ch. 6 - Prob. 6.33SPCh. 6 - What is the difference between a molecule and an...Ch. 6 - Prob. 6.35SPCh. 6 - How many protons and electrons are in each of the...Ch. 6 - What is the identity of the element X in the...Ch. 6 - Prob. 6.38SPCh. 6 - Prob. 6.39SPCh. 6 - Prob. 6.40SPCh. 6 - Prob. 6.41SPCh. 6 - What doubly positive ion has the following...Ch. 6 - Prob. 6.43SPCh. 6 - Prob. 6.44SPCh. 6 - Which element in the transition-metal series Sc...Ch. 6 - Prob. 6.46SPCh. 6 - Prob. 6.47SPCh. 6 - Prob. 6.48SPCh. 6 - Prob. 6.49SPCh. 6 - Prob. 6.50SPCh. 6 - Prob. 6.51SPCh. 6 - Prob. 6.52SPCh. 6 - Prob. 6.53SPCh. 6 - Prob. 6.54SPCh. 6 - Prob. 6.55SPCh. 6 - Prob. 6.56SPCh. 6 - Prob. 6.57SPCh. 6 - Prob. 6.58SPCh. 6 - Prob. 6.59SPCh. 6 - What is the relationship between the electron...Ch. 6 - Prob. 6.61SPCh. 6 - Prob. 6.62SPCh. 6 - Prob. 6.63SPCh. 6 - Prob. 6.64SPCh. 6 - Prob. 6.65SPCh. 6 - Prob. 6.66SPCh. 6 - Prob. 6.67SPCh. 6 - Prob. 6.68SPCh. 6 - Prob. 6.69SPCh. 6 - Prob. 6.70SPCh. 6 - Prob. 6.71SPCh. 6 - Prob. 6.72SPCh. 6 - Prob. 6.73SPCh. 6 - Prob. 6.74SPCh. 6 - Prob. 6.75SPCh. 6 - Find the lattice energy of LiBr(s) in Table 6.3,...Ch. 6 - Prob. 6.77SPCh. 6 - Prob. 6.78SPCh. 6 - Prob. 6.79SPCh. 6 - Prob. 6.80SPCh. 6 - Prob. 6.81SPCh. 6 - Prob. 6.82SPCh. 6 - Prob. 6.83SPCh. 6 - Use the data and the result in Problem 6.78 to...Ch. 6 - Prob. 6.85SPCh. 6 - Prob. 6.86CPCh. 6 - Prob. 6.87CPCh. 6 - Prob. 6.88CPCh. 6 - Prob. 6.89CPCh. 6 - Prob. 6.90CPCh. 6 - Prob. 6.91CPCh. 6 - Prob. 6.92CPCh. 6 - Prob. 6.93CPCh. 6 - Prob. 6.94CPCh. 6 - Prob. 6.95CPCh. 6 - Prob. 6.96CPCh. 6 - Prob. 6.97CPCh. 6 - Prob. 6.98CPCh. 6 - Consider the electronic structure of the element...Ch. 6 - Prob. 6.100MPCh. 6 - Prob. 6.101MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 7.42 Arrange the following in order of increasing first ionization energy: Na, Cl, Al, S, and Cs.arrow_forwardArrange in order of increasing ionization energy. (a) the Group 1A elements H, Li, and Cs (b) the Period 4 elements As, K, & Searrow_forwardIn general, as you go across a period in the periodic table from left to right: (1) the atomic radius (2) the electron affinity becomes (3) the first ionization energy. (1) (2) (3) A decreases, increasingly, increases B. decreases, increasingly, decreases C. increases, increasingly, increases D. increases, increasingly, decreases E. decreases, decreasingly, increases B D E A negative; andarrow_forward
- 7.125. How do ionization energies change with increasing atomic number (a) down a group of elements in the periodic table and (b) from left to right across a period of elements?arrow_forward7.20 Without referring to a periodic table, write the electronconfiguration of elements with the following atomicnumbers: (a) 9, (b) 20, (c) 26, (d) 33.arrow_forward7.89 Which of the following properties show a clear periodicvariation: (a) first ionization energy, (b) molar mass ofthe elements, (c) number of isotopes of an element,(d) atomic radius?arrow_forward
- 7.36 Which is the largest atom in the third period of the periodic table?arrow_forwardRank the elements in each of the following sets in order of increasing atomic radius. (Use the appropriate <, =, or > symbol to separate substances in the list.) (a) Na, Li, H, Rb (b) S, Ar, Si, Alarrow_forward(9) lons and exceptions. Write condensed electron configurations for the following ions and also of elements that do not follow the Periodic Table prediction. Which configuration(s) are/is isoelectronic with neon? Which configuration(s) are/is isoelectronic with that of Argon? (a) 0²- (b) V3+ N3- Ag+ exception (d) (e) Ba²+ (f) U3+ (g) Ni4+ Cr exception (j) CI (a) (c) (e) (g) (1) (c) (h) K+ (1) (b) (d) (f) (i)arrow_forward
- Answer ALL parts of this question. (a) Explain the following observations in first ionisation energies (IE₁). There is a large decrease in IE₁ between He and Li, Ne and Na and, Ar and K. IE₁ is lower for Al than Mg. IE₁ is lower than IE₂. (i) (ii) (iii)arrow_forward6.2) which second row element of the periodic table has the following sequence of successive ionization energies ?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
Atomic Number, Atomic Mass, and the Atomic Structure | How to Pass ChemistryThe Nucleus: Crash Course Chemistry #1; Author: Crash Course;https://www.youtube.com/watch?v=FSyAehMdpyI;License: Standard YouTube License, CC-BY