
Concept explainers
(a)
Interpretation:
By using the given data, high heating value (HHV) vs molar mass of hydrocarbon graph has to be drawn and also the HHV for propane has to be calculated by using that graph.
(a)
Explanation of Solution
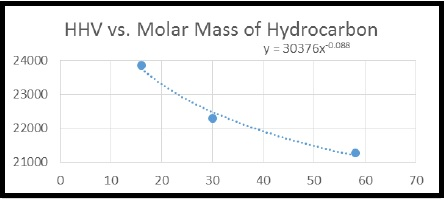
Figure 1
Calculate the HHV for propane by using the trend line equation,
(b)
Interpretation:
By using the given data, High heating value (HHV) - low heating value (LHV) vs molar mass of hydrocarbon graph has to be drawn and also the
(b)
Explanation of Solution
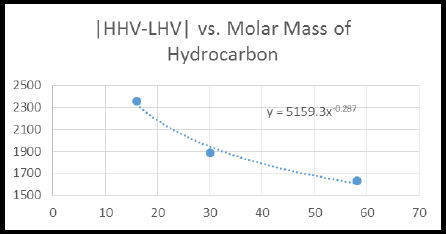
Figure 2
Calculate the
(c)
Interpretation:
By using the given and obtained data in part (a) and part (b), enthalpies of reaction for the given reaction has to be calculated.
(c)
Explanation of Solution
Given reaction
Covert
The second equation corresponds to propane’s LHV. Using
Given reaction
Covert
The estimated enthalpies are
(d)
Interpretation:
By using the given data and results obtained in part (c), the amount of heat energy released when
(d)
Answer to Problem 6.161QP
The amount of heat energy released from the combustion of
Explanation of Solution
Given information,
Use the enthalpy change associated with the LHV based thermochemical equation to calculate the heat energy,
The amount of heat energy released when
(e)
Interpretation:
Estimated results in part (d) has to be compared with accepted thermochemical heats of formation tabulated in appendix C.
Concept introduction:
Hess’s law:
This law is also known as Hess’s law of heat of summation.
This law states that, the sum of enthalpy changes for the individual steps will give the enthalpy change for the overall reaction.
The enthalpy change for the overall chemical change is the same.
(e)
Explanation of Solution
Calculate the enthalpy change using thermochemical heats of formation values
The accepted LHV- based thermochemical equation is
Calculate the heat energy when combustion of
To two significant figures, the obtained value is the same as that calculated in part (d) so the percentage difference is negligible.
Want to see more full solutions like this?
Chapter 6 Solutions
Lab Manual Experiments in General Chemistry
- Suppose that you are studying kinetic energy of helium molecules: A helium weather balloon rises to an altitude of 40,000 ft; the temperature of the gas drops to 70 F. (a) Make an appropriate choice of system and surroundings and describe it unambiguously. (b) Explain why you chose the system and surroundings you did. (c) Identify transfers of energy and material into and out of the system that would be important for you to monitor in your study.arrow_forwardIn 2010, 3.30109 gallons of gasoline were consumed in the United States. The following assumptions can be made: • Gasoline is mainly n-octane, C8H18(d=0.7028g/mL). • Burning one mole of n-octane in oxygen releases 5564.2 kJ of heat. • The heat capacity C of the surface region of the earth is 2.61023 J/K. What is the increase in temperature of the surface region of the earth due to gasoline consumption in the United States?arrow_forward9.102 A runner generates 418 kJ of energy per kilometer from the cellular oxidation of food. The runner's body must dissipate this heat or the body will overheat. Suppose that sweat evaporation is the only important cooling mechanism. If you estimate the enthalpy of evaporation of water as 44 kJ/mol and assume that sweat can he treated as water, describe how you would estimate the volume of sweat that would have to be evaporated if the runner runs a 10-km race.arrow_forward
- Indicate which state function is equal to heat, q, for each process described. a. The ignition of a sample in a bomb calorimeter, an unyielding, heavy metal chamberin which samples are burned for heat content analysis b.The melting of an icecube in a cup c.The cooling down ofthe inside of arefrigerator d.A fire in a fireplacearrow_forwardAn industrial process for manufacturing sulfuric acid, H2SO4, uses hydrogen sulfide, H2S, from the purification of natural gas. In the first step of this process, the hydrogen sulfide is burned to obtain sulfur dioxide, SO2. 2H2S(g)+3O2(g)2H2O(l)+2SO2(g);H=1124kJ The density of sulfur dioxide at 25C and 1.00 atm is 2.62 g/L, and the molar heat capacity is 30.2 J/(mol C). (a) How much heat would be evolved in producing 1.00 L of SO2 at 25C and 1.00 atm? (b) Suppose heat from this reaction is used to heat 1.00 L of the SO2 from 25C to 500C for its use in the next step of the process. What percentage of the heat evolved is required for this?arrow_forwardYou did an experiment in which you found that 59.8 J was required to raise the temperature of 25.0 g of ethylene glycol (a compound used as antifreeze in automobile engines) by 1.00 K. Calculate the specific heat capacity of ethylene glycol from these data.arrow_forward
- The enthalpy change for the following reaction is 393.5 kJ. C(s,graphite)+O2(g)CO2(g) (a) Is energy released from or absorbed by the system in this reaction? (b) What quantities of reactants and products are assumed? (c) Predict the enthalpy change observed when 3.00 g carbon burns in an excess of oxygen.arrow_forwardNitrogen gas (2.75 L) is confined in a cylinder under constant atmospheric pressure (1.01 105 pascals). The volume of gas decreases to 2.10 L when 485 J of energy is transferred as heat to the surroundings. What is the change in internal energy of the gas?arrow_forwardGasohol, a mixture of gasoline and ethanol, C2H5OH, is used as automobile fuel. The alcohol releases energy in a combustion reaction with O2. C2H5OH(l)+3O2(g)2CO2(g)+3H2O(l) If 0.115 g ethanol evolves 3.62 kJ when burned at constant pressure, calculate the combustion enthalpy for ethanol.arrow_forward
- In a coffee-cup calorimeter, 1.60 g NH4NO3 is mixed with 75.0 g water at an initial temperature of 25.00C. After dissolution of the salt, the final temperature of the calorimeter contents is 23.34C. Assuming the solution has a heat capacity of 4.18 J/C g and assuming no heat loss to the calorimeter, calculate the enthalpy change for the dissolution of NH4NO3 in units of kJ/mol.arrow_forwardsample of natural gas is analyzed and found to be88.4% methane (CH4) and 11.6% ethane (C2H6) bymass. The standard enthalpy of combustion of methaneto gaseous carbon dioxide (CO2) and liquid water (H2O) is -891 kJ/mol. Write the equation for the combustionof gaseous ethane to carbon dioxide and water.Calculate the standard enthalpy of combustion of ethaneusing standard enthalpies of formation from Table R-11on page 975. Using that result and the standard enthalpyof combustion of methane in Table 15.3, calculate theenergy released by the combustion of 1 kg of natural gas.arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





