
Concept explainers
Drawing Structural and Condensed Structural Formulas
Give the molecular formula, and draw the structural formula and condensed structural formula for the
Interpretation:
The molecular formula, structural formula and condensed structural formula are to be represented for the alkane with
Concept Introduction:
The organic compounds which consist of only carbon and hydrogen atoms are called hydrocarbons. The atoms in hydrocarbons are linked by single, double, or triple bonds.
The graphic representation of a chemical compound is called its structural formula. It represents the arrangement of atoms.
In a condensed structural formula, the bond between atoms in a compound is represented by the absence of a line.
The chemical formula that represents the total number of atoms arranged in a molecule is known as the molecular formula.
Answer to Problem 6.1YT
Solution: Molecular formula for an alkane is given as:
The structural formula for an alkane contains
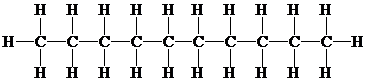
The condensed structural formula for an alkane has
Or
Explanation of Solution
The molecular formula for an alkane with
The structural formula for an alkane compound which contains
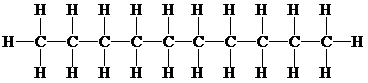
The condensed structural formula for a pentane consists of
Or
Molecular formula, the structural formula and the condensed structural formula for an alkane has
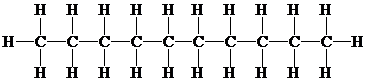
and
Or
Want to see more full solutions like this?
Chapter 6 Solutions
Bundle: Chemistry In Focus: A Molecular View Of Our World, 6th + Owlv2 6-month Printed Access Card
- Is the general formula of a cycloalkanes the same as the general formula of an alkane, CnH2n+2? Draw any structural diagram to illustrate your answer.arrow_forwardthe mass of an unknown volume of air is equal to 0.123 grams, another alkane with the same volume as air has a mass of 0.246. a) Determine the molecular formula of the alkane and the complete combustion reaction of the complete alkane. b) find the volume of gases formed under normal conditions.arrow_forwardDraw the following organic compound (condensed formula) isopropyl bromidearrow_forward
- Draw and name all of the structural isomers of a) pentane and b) hexane.arrow_forwardWrite the chemical reaction involved in the reactions of hydrocarbon. Experiment: Solubility test (drops of a dye was added to each test tube containing the test sample) Substances: (a) Cyclohexane , (b) Cyclohexene , (c) Toluenearrow_forwardIn each of the following classes of organic compounds, write a molecule with a total of 16 carbons (by express formula) with at least one aromatic group and one alkyl group in its structure. Name each structure you write according to the IUPAC system. a) Write down and name a dithiol molecule. b) Write down a lactone molecule and name it.arrow_forward
- 1. The torch used to start the modern Olympic Games uses a mixture ofpropane and butane. When propane and butane burn in the air, they produce heat, energy, and a flame. A) Propane and butane are chemical compounds. Name the two elements that make these compounds. B) State the name of the compounds that contain the elements in part A C) To which homologous series do propane and butane belong?arrow_forwardQuestions On complete combustion, one mole of an alkane produces two moles of carbon dioxide and three moles of water. Suggest the molecular formula of this alkane. 1.arrow_forwardAn organic compound (A) has a general formula CnH2n. This compound has five carbon atoms. When the compound is hydrogenated, it forms a compound (B) having two excess H atoms. a) Identify the compound (A), write its name. b) Identify the compound (B), write its name. c) Which out of (A) and (B) is a saturated hydrocarbon? d) Which of the following figures can be regarded as an Isomer of (B)? ye-cny -CHg-CH-Cly Figure- C43 Fgurea C-2H-cr-cly yc--cns HC igurearrow_forward
- c. Write the condensed structural formula for each of the following moleculesarrow_forwardQuestion 18 of 23 Provide the correct IUPAC name for the skeletal (line-bond) structure shown here.arrow_forwardThe general formula of an alkane is CnH2n+2 . What is the general formula of an (a) alkene? (b) alkyne? (c) alcohol derived from an alkane?arrow_forward

 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





