(a)
Interpretation:
The systematic name and the chemical formula for plumbic fluoride have to be written.
Concept Introduction:
In the older nomenclature system, an “–ous” ending is used to indicate the lower common ionic charge, and an “–ic” ending is used to indicate the higher common ionic charge.
Some of the examples are shown below,
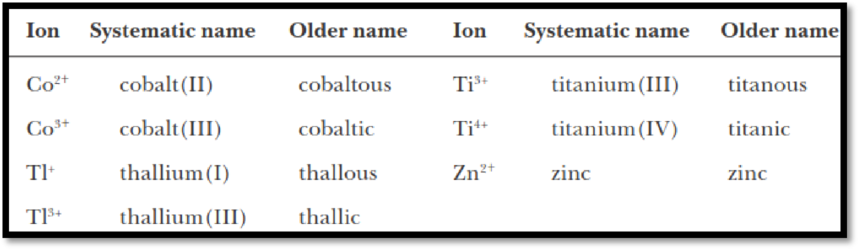
Figure 1
Chemical formula:
- 1) The chemical formula of binary compound from a pair of its constituent ion has to be written.
- 2) The coefficients of the ions with the magnitude of the charge on each of the ions have to be crossed multiplied. This is done to neutralize charge on the binary compound.
- 3) The oxidation state of the metal is written in roman numbers in the brackets
(b)
Interpretation:
The systematic name and the chemical formula for mercurous chloride have to be written.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The systematic name and the chemical formula for plumbous sulphide have to be written.
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The systematic name and the chemical formula for mercuric oxide have to be written.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Student Solutions Manual to Accompany General Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





