
(a)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanamine (
Concept introduction:
Leveling effect refers to the effect of a solvent on the properties of acids and bases. For an acid-base reaction, the strength of the strong acid is limited or leveled by the basicity of the solvent. Similarly, the strength of the strong base is leveled by the acidity of the solvent. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.46P
With respect to the leveling effect, ethanamine is a suitable solvent for a reaction involving the chloride ion
Explanation of Solution
The reaction of chloride ion
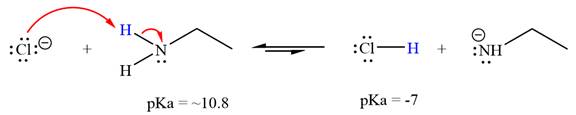
Hydrochloric acid,
The solvent effect on the reactant is determined with respect to the leveling effect.
(b)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanamine (
Concept introduction:
Leveling effect refers to the effect of a solvent on the properties of acids and bases. For an acid-base reaction, the strength of the strong acid is limited or leveled by the basicity of the solvent. Similarly, the strength of the strong base is leveled by the acidity of the solvent. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.46P
With respect to the leveling effect, ethanamine is a not suitable solvent for a reaction involving
Explanation of Solution
The reaction of
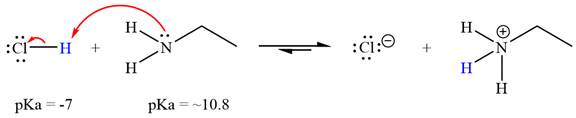
Hydrochloric acid,
The solvent effect on the reactant is determined with respect to the leveling effect.
(c)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanamine (
Concept introduction:
Leveling effect refers to the effect of a solvent on the properties of acids and bases. For an acid-base reaction, the strength of the strong acid is limited or leveled by the basicity of the solvent. Similarly, the strength of the strong base is leveled by the acidity of the solvent. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.46P
With respect to the leveling effect, ethanamine is not a suitable solvent for a reaction involving
Explanation of Solution
The reaction of

Ethanamine,
The solvent effect on the reactant is determined with respect to the leveling effect.
(d)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanamine (
Concept introduction:
Leveling effect refers to the effect of a solvent on the properties of acids and bases. For an acid-base reaction, the strength of the strong acid is limited or leveled by the basicity of the solvent. Similarly, the strength of the strong base is leveled by the acidity of the solvent. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.46P
With respect to the leveling effect, ethanamine is not a suitable solvent for a reaction involving
Explanation of Solution
The reaction of
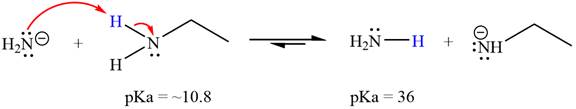
Ethanamine,
The solvent effect on the reactant is determined with respect to the leveling effect.
(e)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanamine (
Concept introduction:
Leveling effect refers to the effect of a solvent on the properties of acids and bases. For an acid-base reaction, the strength of the strong acid is limited or leveled by the basicity of the solvent. Similarly, the strength of the strong base is leveled by the acidity of the solvent. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.46P
With respect to the leveling effect, ethanamine is not a suitable solvent for a reaction involving
Explanation of Solution
The reaction of
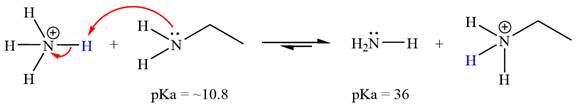
Ethanamine,
The solvent effect on the reactant is determined with respect to the leveling effect.
(f)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanamine ( ) as a solvent with respect to leveling effect.
Concept introduction:
Leveling effect refers to the effect of a solvent on the properties of acids and bases. For an acid-base reaction, the strength of the strong acid is limited or leveled by the basicity of the solvent. Similarly, the strength of the strong base is leveled by the acidity of the solvent. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.46P
With respect to the leveling effect, ethanamine is not a suitable solvent for a reaction involving
Explanation of Solution
The reaction of hydroxide ion
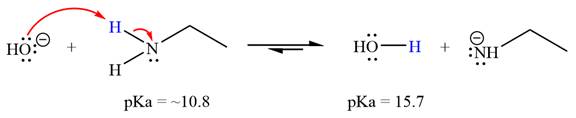
Ethanamine,
The solvent effect on the reactant is determined with respect to the leveling effect.
Want to see more full solutions like this?
Chapter 6 Solutions
ORGANIC CHEMISTRY PRINCIPLES & MECHANISM
- Formaldehyde, H2C=O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no carbonyl groups. Only one monobromo derivative (C3H5BrO3) of trioxane is possible. Propose a structure for trioxane.arrow_forwardBourgeonal is an aldehyde with the odor of the Lily of the Valley. If Bourgeonal has M^+=190 and contains 4 double bond(s) and 1 ring(s); what is its molecular formula?arrow_forwardAn unknown hydrocarbon Q has a formula C6H12. Q Reacts with osmium tetroxide to give a diol R. When oxidized with KMnO4 in an acidic medium, Q gives two products. One product is propanoic acid and the other a ketone S. Provide reaction equations to identify the possible structures of Q, R and S.arrow_forward
- The five parts of question 16 relate to the following three molecules: N-H А в с A (a) Which of the three molecules above are structural isomers? (b) Which of the molecules contains a carbon with linear geometry? (c) Which molecule contains a tertiary amine? (d) Which molecule only contains sp³ hybridized atoms (not including hydrogen)? (e) Which molecule is chiral? Circle the stereocentre in this molecule.arrow_forwarda Complete the following reaction of a disulfide with a reducing agent. What is the structure of the organic product (of which two moles are formed)? CH3CH2-S-S-CH2CH3 + 2(H) · C P орy aste CH4 ChemDoodle®arrow_forward1: In the sweetening MEROX process for kerosene the mercaptans are converted into (a) Sulpher (b) Disulphide (c) Hydrogen sulphide (d) None of these 2: Which of the following products contain maximum sulphur? (a) Diesel fuel (b) Fuel oil (c) Jet fuel (d) LPG 3: Which of the following petroleum product has a maximum C/H ratio (by weight)? (a) Light diesel oil (b) Fuel oil (c) Naphtha (d) Heating oil 4: Which of the following hydrocarbon are most desirable in gasoline? (a) Paraffins (b) Isoparaffins (c) Naphthenes (d) Aromatic 5: Octane no. of paraffins: (a) Remain constant with change in the number of carbon atoms (b) Increases with increase in the number of carbon atoms (c)Decreases with increase in the number of carbon atoms (d)None of the above 6: Which of the following hydrocarbons has a maximum octane number? (a) Benzene (b) Cyclohexane (c) Hexane (d) Iso-hexane 7: Which of the following petroleum products has a minimum flashpoint? (a)Gasoline (b) Kerosene (c) Fuel oil (d) Heating…arrow_forward
- 4 Determine how many carbon environments each of the following molecules posess.arrow_forwardDraw the structure for the following compounds:(i) Ethanoic acid(ii) Bromopentane(iii) Butane(iv) Hexanalarrow_forwardHydrogen peroxide is not only an antiseptic but also an oxidizing agent. The following equation shows the reaction of hydrogen peroxide with acetaldehyde to give acetic acid: C2H4O(l) + H2O2(l) → C2H4O2(l) + H2O(l) In this reaction, which species is oxidized and which species is reduced? Which species is the oxidizing agent and which species is the reducing agent?arrow_forward
- Diazomethane, CH2N2, is used in the organic chemistry laboratory despite its danger because it produces very high yields and is selective for reaction with carboxylic acids. Write the products of the following reactions.arrow_forwardTreating choline with acetic anhydride gives acetylcholine, a neurotransmitter. Write an equation for the formation of acetylcholine. (CH,),NCH,CH,OH Cholinearrow_forwardProvide the major organic products for the followingarrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

