
Concept explainers
(a)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, σ bond or the region where the bond is formed if the new bond is a π bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
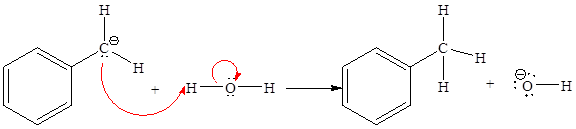
Explanation of Solution
The given proton transfer reaction is
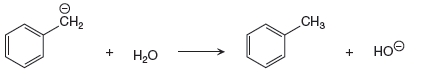
In the above reaction, the bond
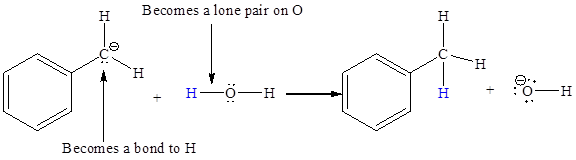
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on C to the H on water (highlighted blue) to illustrate the formation of
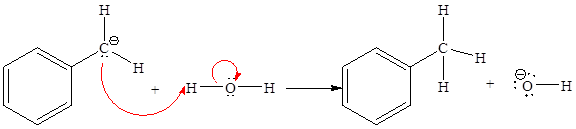
The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
(b)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, ![]() bond or the region where the bond is formed if the new bond is a
bond or the region where the bond is formed if the new bond is a ![]() bond.
bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
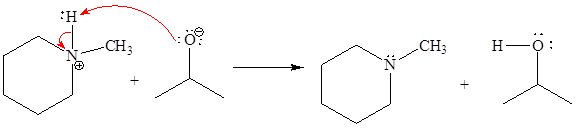
Explanation of Solution
The given proton transfer reaction is
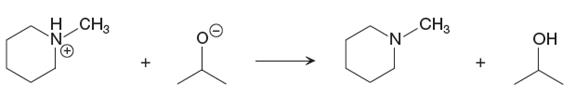
In the above reaction, the bond
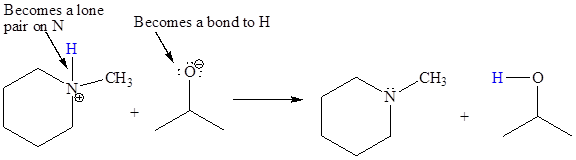
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on O to the H on N (highlighted blue) to illustrate the formation of the

The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
(c)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, ![]() bond or the region where the bond is formed if the new bond is a
bond or the region where the bond is formed if the new bond is a ![]() bond.
bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
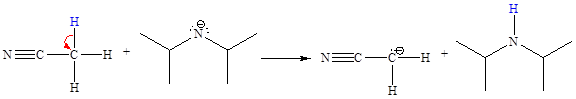
Explanation of Solution
The given proton transfer reaction is
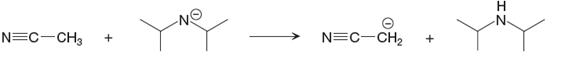
In the above reaction, the bond
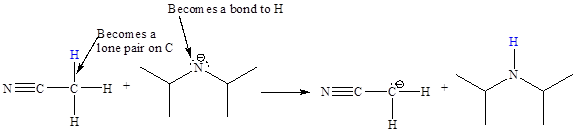
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on N to the H on C (highlighted blue) to illustrate the formation of the
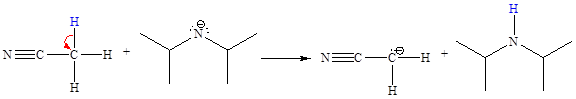
The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
(d)
Interpretation:
Missing curved arrows are to be supplied for the given proton transfer reaction. The relevant electrons are to be drawn if they are not shown.
Concept introduction:
In a proton transfer reaction, a proton is transferred from a Bronsted-Lowry acid to a Bronsted-Lowry base in a single elementary step in which one bond is broken and another is formed simultaneously. The curved arrow notation shows the movement of valence electrons, not atoms. The movement of two electrons is shown be using a double-barbed arrow. To represent bond breaking, the tail of the arrow originates from the center of a bond whereas to represent bond formation, the head of arrow points to an atom which forms the new bond, that is, ![]() bond or the region where the bond is formed if the new bond is a
bond or the region where the bond is formed if the new bond is a ![]() bond.
bond.
Answer to Problem 6.39P
The missing curved arrow notation for the proton transfer reaction and relevant electrons is shown as
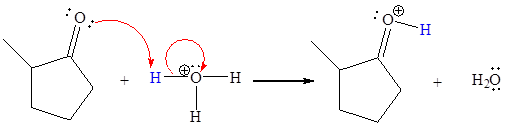
Explanation of Solution
The given proton transfer reaction is
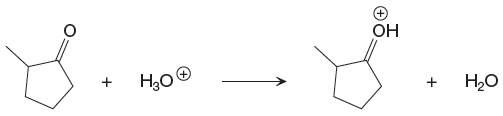
In the above reaction, the bond
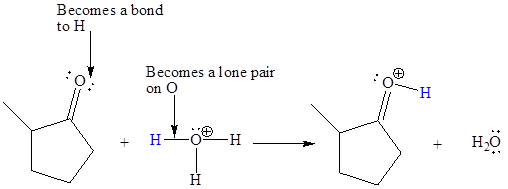
The appropriate movement of these valence electrons is shown by using curved arrow notations. One curved arrow is to be drawn from the lone pair on O to the H of
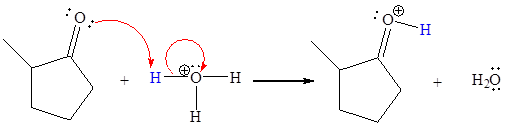
The curved arrow notation for the proton transfer of the given reaction is drawn on the basis of the movement of valence electrons involved in bond breaking and bond formation.
Want to see more full solutions like this?
Chapter 6 Solutions
ORGANIC CHEMISTRY PRINCIPLES & MECHANISM
- Label the acid and base, and the conjugate acid and base, in the following reaction. Use curved arrow notation to show the movement of electron pairs. (See the Attachment)arrow_forwardCan you explain all the steps to get the answer? And how do you know the equilibrium? What does the arrows mean? For the given acid base reaction: a) Determine which is the weakest (most stable) base Show the details for your reasoning. b) Show which side of the reaction is favored at equilibrium and explain why. c) Draw the curved arrows to show the proton transfer from the acid to the base the given acid-base reaction: a) Determine which is the weakest (most stable) base. Show the details for your reasoning. b) Show which side of the reaction is favored at equilibrium and explain why c) Draw the curved arrows to show the proton transfer from the acid to the base.arrow_forwardDo all label answer to which so I know the table is there to help answer questions if neededarrow_forward
- (Intermediate) Reactant (Intermediate) Product H3C 'o: Apply Mechanism Hint Solution 2 remaining step(s) can be solved Tip: Only add curved arrows in this sketcherarrow_forwardThe following are equivalent ways of asking about the acidity of an H atom: • What is the most acidic H on the molecule? • Which H is associated with the published pKa value? • Which H on the molecule is easiest to remove? • Which H on the molecule takes the least energy to remove? • Which bond to an H is most polarized? • For which H atom is removal least uphill in energy? • Which bond to an H atom, when broken, results in the lowest PE conjugate base? We will often find the last of these questions is easiest to answer. To do this, find all the different Hatoms on the molecule, and draw all possible conjugate bases.Only the lowest-energy one is the “real” conjugate base. Identify this structure, and you have found the most acidic H. Use this strategy to find the most acidic H on each of the following molecules. Note: Each structure hasat least three different kinds of H’s, so draw at least three unique conjugate bases for each.arrow_forwardSummarize the relationship between pKa and base strength by completing the followingsentences: a. For a given base, the higher the pKa of its conjugate acid, the stronger or weaker the base. b. For a given base, the lower the pKa of its conjugate acid, the stronger or weaker the base.arrow_forward
- 2. The following pair of compounds can undergo an acid/base reaction. a) Identify the base and the acid b) Draw the products of the reaction and a mechanism for the proton transfer. c) Based on qualitative concepts, is this reaction favored? Explain. Siarrow_forwardAmong three bases, X, Y¯, and Z¯, the strongest one is Y¯, and the weakest one is Z-. Rank their conjugate acids, HX, HY, and HZ, in order of decreasing strength. Rank the acids from strongest to weakest. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Help HZ HY HX Strongest acid Weakest acidarrow_forwardHi , can you help me to answer this question .. can you draw curved arrows to move a proton from acid to the base .. Identify acid , base , conjugate acid and conjugate base also draw the product of proton transfer.. I need the full scheme answe so that i can understand this topic better.. Thank youuarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning


