
Concept explainers
The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism.
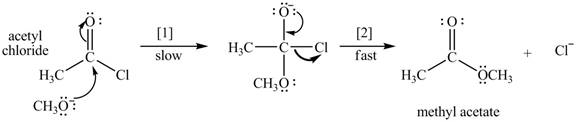
a. Write the rate equation for this reaction, assuming the first step is rate-determining.
b. If the concentration of
c. If the concentrations of both
d. Classify the conversion of acetyl chloride to methyl acetate as an addition, elimination, or substitution.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry-Package(Custom)
- Consider the following energy plots for a chemical reaction when answering the questions below. a. Which plot (purple or blue) is the catalyzed pathway? How do you know? b. What does E1 represent? c. What does E2 represent? d. Is the reaction endothermic or exothermic?arrow_forwarda) Sketch the energy diagram for the conversion of tertiary butyl bromide to methyl t-butyl ether. Note that this is a two step reaction with the t-butyl cation as an intermediate. Assume that this is overall an exothermic reaction. Show the location of both transition states and the intermediate. The first step has the higher energy of activation and is the rate determining step. (CH3)3C-Br ------> (CH3)3C+ + Br- (CH3)3C+ + H3CO- ------> (CH3)3C-O-CH3 (b)Draw the curved arrow mechanism and the transition state when the tertiary butyl carbocation reacts with the methoxide ion to give an ether. (CH3)3C+ + H3CO- ------> (CH3)3C-O-CH3arrow_forwardFor the reaction CH 3 Br (aq) + OH - (aq) → CH 3 OH (aq) + Br- (aq) it is known that when the concentration of OH - doubles, the reaction rate doubles. When the concentration of CH 3 Br increases 1.2 times, the reaction rate increases 1.2 times. Write the rate equation for the reactionarrow_forward
- 1. A chemist wishes to determine the rate of reaction of zinc with hydrochloric acid. The equation for the reaction is: Zn (s) + 2HCl (aq) → H2 (g) + ZnCl2 (aq) It is determined that the rate of Zn consumption is -0.00040 mol/s. What would be the rate of HCl consumption Answer to 6 decimal places 2. A chemist wishes to determine the rate of reaction of zinc with hydrochloric acid. The equation for the reaction is: Zn (s) + 2HCl (aq) → H2 (g) + ZnCl2 (aq) If initially there was 0.00400 moles of Zn and after 17 s there was 0.00160 moles of zinc, what is the rate of reaction in moles of Zn consumed per second? Answer to 6 decimal places 3. For the reaction C6H12 + 9 O2→6 CO2 + 6 H2O, select all the correct relations of rate. Question 3 options: 16∆[CO2]∆t 16∆[H2O]∆t -∆[C6H12]∆t 4. For the reaction 3 H2 + N2 → 2 NH3, what is the rate of reaction when 0.062 moles of NH3 at 6.0 s then becomes 0.190 moles of…arrow_forward1) If the reaction order of reactant A is 1, and the reaction order of reactant B is 2, what is the overall reaction order? 2) How is the value of the rate constant determined? 3) How does the value of the rate constant effect the reaction rate?arrow_forwardComplete the following reaction by filling in the blank 165 Yb + ____ ------> ____ + ?e options : 165Tm 165 Er n p 165 Lu e+ 161 Er e- 165 Hfarrow_forward
- When a small amount of iodine is added to a mixture of chlorine and methane, it prevents chlorination from occurring. Therefore, iodine is a free-radical inhibitor for this reaction. Calculate ∆H° values for the possible reactions of iodine with species present in the chlorination of methane, and use these values to explain why iodine inhibits the reaction. (The I¬Cl bond-dissociation enthalpy is 211 kJ>mol or 50 kcal>mol.)arrow_forward1. Write the mechanism for the bromination of ethane.2. What simple chemical tests may be used to differentiate the alkene from an alkane? Describe the test.arrow_forwardProduct X contains a photosensitive reagent A in first order. The scientist mistakenly took out Product X in a transparent bottle, exposing it to sunlight. Determine the time (mins) it takes to decrease the concentration of A in half, given the rate constant equal to 7.49 x 10-4/s. Please use 2 decimal places. Do NOT include units in your final answer.arrow_forward
- Average reaction rate between 0 and 100 us? Average reaction rate between 200 and 300 us?arrow_forward1. base on the data,what how do we find rate constant for this reaction at room temperature? 2. what cause the formation of the purple color ??arrow_forwardWhy does adding the reaction mix to the “stop” tubes immediately stop the enzymic reaction?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





