
(a)
Interpretation:
Structure of the missing substance that involves in the given reaction has to be drawn.
Concept Introduction:
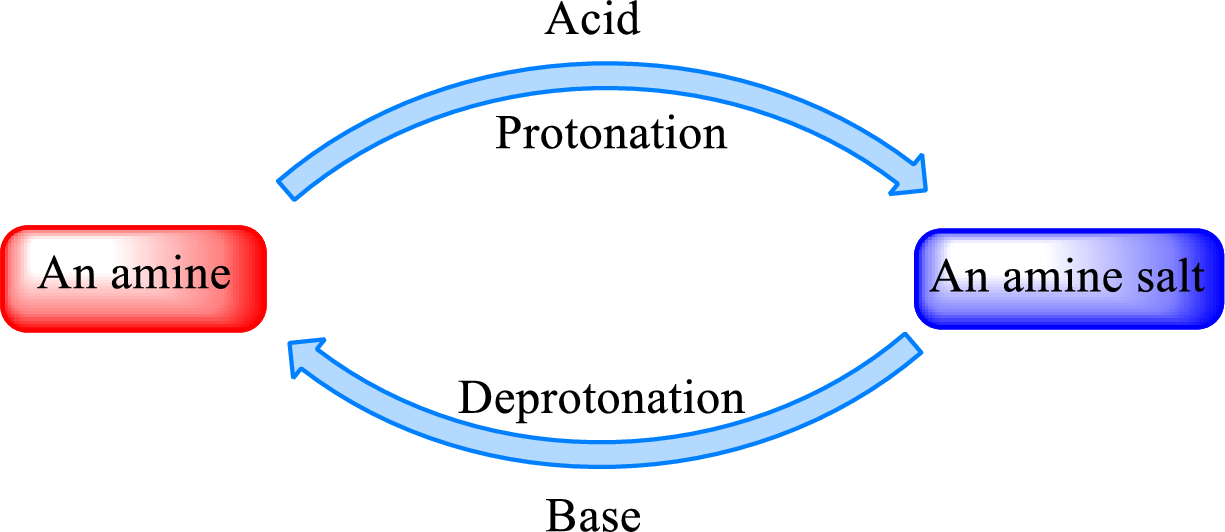
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
(b)
Interpretation:
Structure of the missing substance that involves in the given reaction has to be drawn.
Concept Introduction:
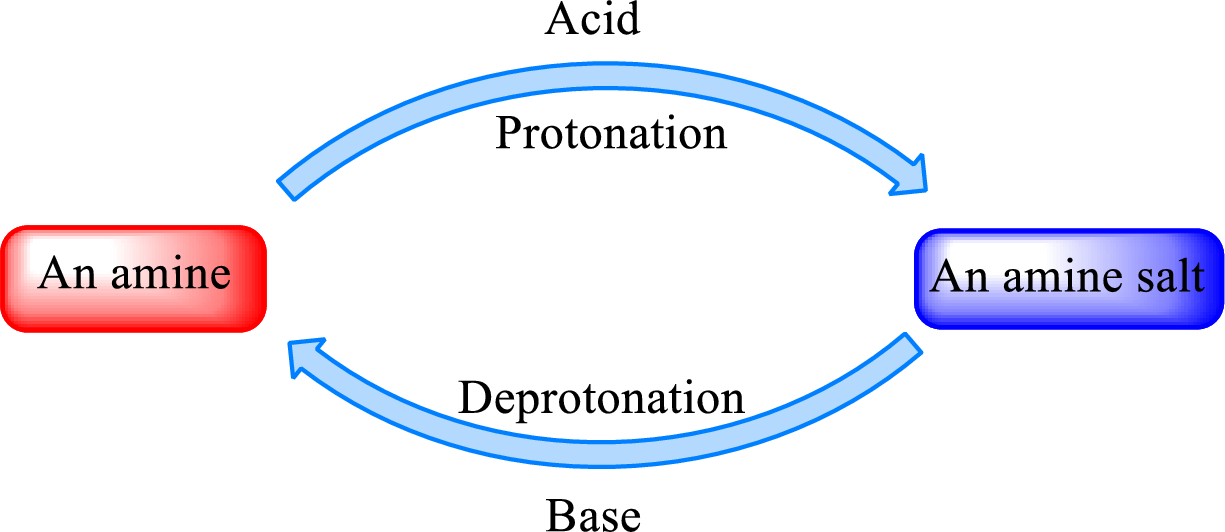
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As amines are bases due to the amino group in it, the reaction with inorganic acid or carboxylic acid gives salt as product. The salt formed is an amine salt. Proton is donated from the acid to the nitrogen atom which acts as a proton acceptor. In simple words, it can be said that in an amine‑acid reaction, the acid loses a hydrogen ion and amine gains a hydrogen ion.
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
(c)
Interpretation:
Structure of the missing substance that involves in the given reaction has to be drawn.
Concept Introduction:
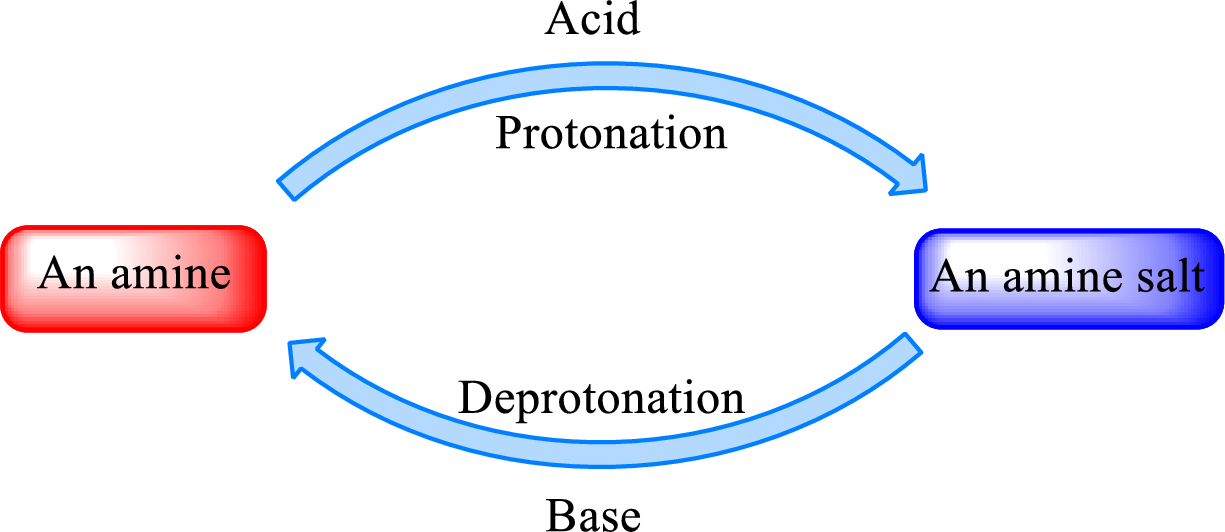
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As amines are bases due to the amino group in it, the reaction with inorganic acid or carboxylic acid gives salt as product. The salt formed is an amine salt. Proton is donated from the acid to the nitrogen atom which acts as a proton acceptor. In simple words, it can be said that in an amine‑acid reaction, the acid loses a hydrogen ion and amine gains a hydrogen ion.
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
(d)
Interpretation:
Structure of the missing substance that involves in the given reaction has to be drawn.
Concept Introduction:
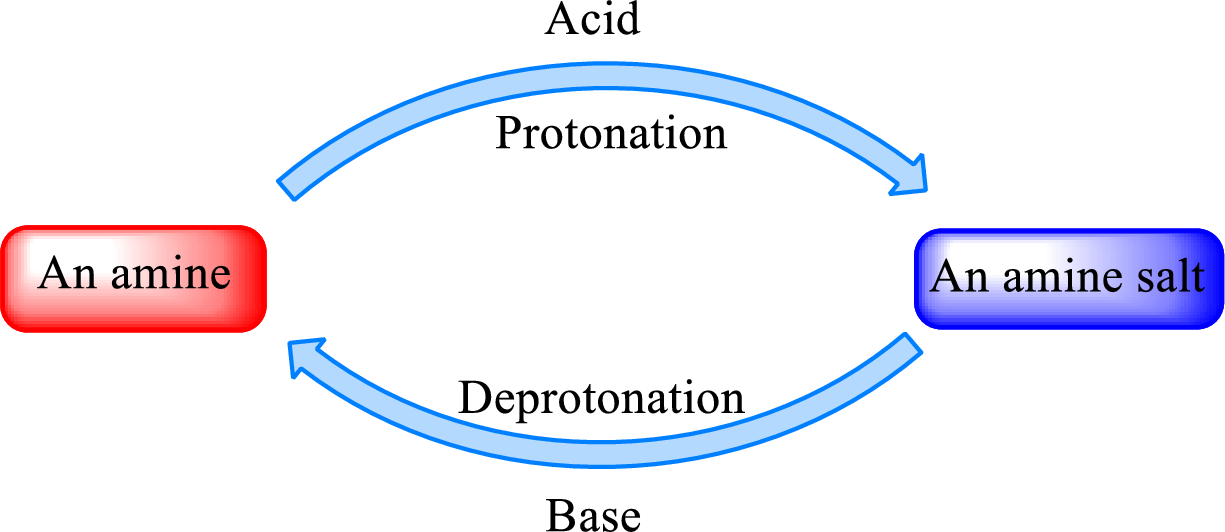
Neutralization reaction is the one that takes place between an acid and a base to give salt as product. As amines are bases due to the amino group in it, the reaction with inorganic acid or carboxylic acid gives salt as product. The salt formed is an amine salt. Proton is donated from the acid to the nitrogen atom which acts as a proton acceptor. In simple words, it can be said that in an amine‑acid reaction, the acid loses a hydrogen ion and amine gains a hydrogen ion.
When a strong base is added to the amine salt, the parent amine can be obtained. This is a reverse reaction of the amine salt formation reaction. These reactions can be represented as shown below,
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic And Biological Chemistry
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


