
Concept explainers
Interpretation:
A reaction roadmap have to be made for the reactions in the study Guide section of chapters 6-11.
Concept Introduction:
Markovnikov addition: The addition reaction of parotic acids to a different alkene or alkyne, the hydrogen atom of
Anti-Markovnikov addition: These rules describe the regiochemistry where the substituent is bonded to a less substituted carbon, rather than the more substituted carbon. This placed is quite unusual as carbon cations which are commonly formed during alkene or alkyne reactions tend to favor the more substituted carbon.
Unsaturated compound: The nucleophile reacts with
The alkylation at the β carbon of ketone or aldehyde is done by the following mechanism;
(1) Alkylating the β carbon via enamine intermediate.
(2) Alkylating the β carbon via a Michael reaction.
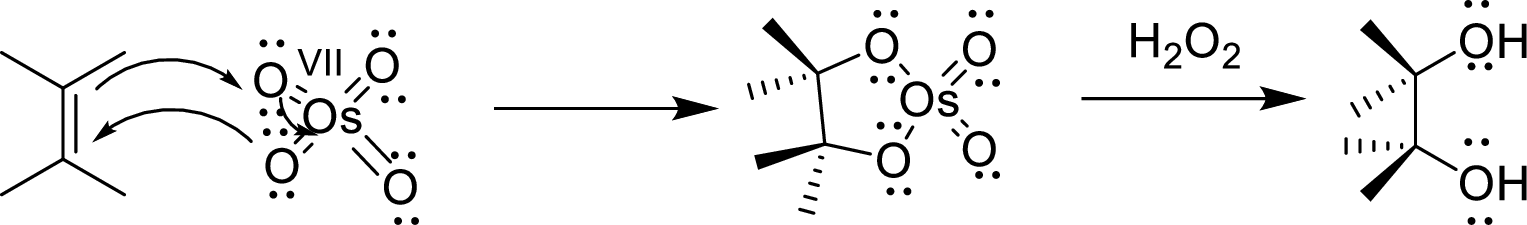
Osmium tetroxide
The reaction mechanism involves the concentrated addition of the osmium tetroxide to the
Lindlar catalyst: The catalyst is used for the hydrogenation of
Ozonolysis Reaction: It is an oxidative reaction which is used to oxidize the carbon-carbon double and triple bond.
Trending nowThis is a popular solution!

Chapter 6 Solutions
Organic Chemistry
- We now continue the use of organic chemistry reaction roadmaps. Because of the unique nature of the new reactions presented, we recommend that you make a new roadmap only for Chapters 2023. To make your own roadmap for Chapters 2023, take a blank sheet of paper and write the following functional groups in the orientations shown. Fill the entire sheet of paper and leave plenty of room between functional groups. Most students find it helpful to use a poster-sized sheet of paper filled out in landscape orientation. We now continue the use of organic chemistry reaction roadmaps. Because of the unique nature of the new reactions presented, we recommend that you make a new roadmap only for Chapters 2023. To make your own roadmap for Chapters 2023, take a blank sheet of paper and write the following functional groups in the orientations shown. Fill the entire sheet of paper and leave plenty of room between functional groups. Most students find it helpful to use a poster-sized sheet of paper filled out in landscape orientation.arrow_forwardIn each of the following classes of organic compounds, write (by express formula) two molecules each with at least one aromatic group and one alkyl group with a total of 10 carbons. Name each structure you write according to the IUPAC system. a)Write and name two diazonium salt. b)Write and name two 2,3-unsaturated aldehyde molecules. c)Write and name two corboxylic acid anhydride molecules.arrow_forwardPropose how the following molecules can be synthesized from the illustrated starting material. Draw all reactants, reagents, catalysts, and other components necessary for each step. Please give simple steps learned in first year and second year organic chemistryarrow_forward
- Consider compound I below, which is structurally related to a natural product that was isolated from an extract of a Caribbean marine sponge (See J. Chem. Soc. 1994, 116, 6015). Answer the following questions about this compound ( Please Explain Answers) How primary alcohols are present? _________ How many secondary alcohols are present? _________ How many quaternary carbons are present? _______arrow_forwardThis question concerns adrenalin which is a hormone, a chemical messenger which stimulates the central nervous system. It's structure is shown below. It has a number of functional groups. What is a functional group? Identify and name the functional groups. Give the reagents and reaction conditions that establish the presence of any TWO of the functional groups present in adrenalinarrow_forwardOrganic Chemistry Acid catalyzed dehydration reaction of 2-methyl-1-butanol produces 2-methyl-2-butene as the major product. Also acid catalyzed dehydration reaction of 3-methyl-1-butanol give the same product as major product. Explain the reason why both of the reaction produce the same product as the major product.arrow_forward
- In each of the organic compound classes given below, write a molecule with a total of 16 carbons (by express formula), with at least one aromatic group and one alkyl group in its structure. Name each structure you write according to the IUPAC system.i) Write and name a molecule of corboxylated acid anhydride.j) Write down and name a half ketal molecule.k) Write and name an N, N-diarylamide molecule.arrow_forwardOrganic Chemistry Incorrect Separation Scheme Cannot be hand-drawn *see attached for the incorrect separation scheme provided. The top of the separation scheme shows what other compound is mixed with your molecule (2,6-dimethyloct-2-ene). Assume for the purposes of this assignment that both compounds are solid at room temperature. Also assume that both compounds are soluble in ether, except ionic compounds. The goal of the separation is the isolate each of the two compounds from the mixture. Below the incorrect separation scheme write a discussion of this incorrect scheme identifying all of the mistakes in the separation scheme. Keep in mind that there will be more than one mistake in the scheme. For each mistake, give a detailed, scientific explanation of why it is incorrect.arrow_forwardI have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:arrow_forward
- What is the natural source of jasmonic acid in detail according to organic chemistry?arrow_forwardIn each of the following classes of organic compounds, write a molecule with a total of 16 carbons (by express formula) with at least one aromatic group and one alkyl group in its structure. Name each structure you write according to the IUPAC system.a) Write and name a fused ring aromatic molecule.b) Write and name the molecule like a meta-disubstitution.c) Write down and name an alkynylphenol molecule.d) Write and name an aromatic dinitro molecule.e) Write and name a heteroaromatic molecule.f) Write and name a benzaldehyde derivative.g) Write down and name a ditivol molecule.h) Write and name a lactone molecule.arrow_forwardCompound A and compound B are in equilibrium. Write a stepwise mechanism from compound Ato compound B showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and products are formed. Show all lone pairs and formal charges. Lastly, explain which compound (Aor B) will be in higher concentration.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

