
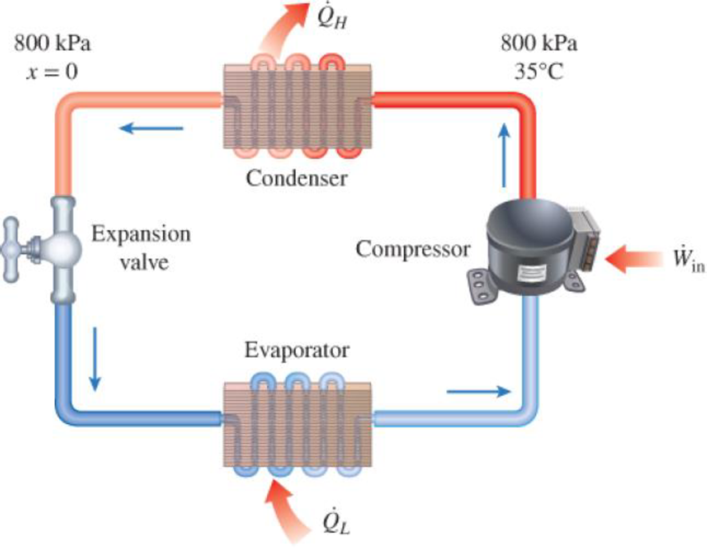
Refrigerant-134a enters the condenser of a residential heat pump at 800 kPa and 35°C at a rate of 0.018 kg/s and leaves at 800 kPa as a saturated liquid. If the compressor consumes 1.2 kW of power, determine (a) the COP of the heat pump and (b) the rate of heat absorption from the outside air.
FIGURE P6–57
(a)
The COP of the heat pump.
Answer to Problem 55P
The COP of the heat pump is
Explanation of Solution
Write the expression for the energy balance equation.
Here, the total energy entering the system is
Simplify Equation (II) and write energy balance relation of refrigrent-134a.
Here, the rate of work to be done into the system is
Substitute
Write the expression for the rate of coefficient performance of a heat pump.
Here the rate of required input of the heat pump is
Conclusion:
Convert the unit of pressure from kPa to MPa.
Refer to Table A-13, “Superheated refrigerant-134a”, obtain the below properties at the superheated pressure and temperature of 800 kPa (0.80 MPa) and 35 C using interpolation method of two variables.
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y are superheated temperature and specific enthalpy.
Show the temperature at 31.31 C and 40 C as in Table (1).
| Temperature, C | Specific enthaply, |
| Saturated liquid, | |
| 31.31 C | 267.34 |
| 35 C | ? |
| 40 C | 276.46 |
Calculate superheated pressure and temperature of 800 kPa (0.80 MPa) and 35 C for liquid phase using interpolation method.
Substitute 31.31 C for
From above calculation the initial enthalpy of condenser is
Refer to Table A-12, “Saturated pressure table” obtain properties at the superheated pressure and quality of final state of 800 kPa and 0.
Write the expression of final specific enthalpy of a two-phase system for condenser.
Here, the specific enthalpy of condenser is
Substitute
Substitute
Substitute
Thus, the COP of the heat pump is
(b)
The rate of heat absorbed from the outside air.
Answer to Problem 55P
The rate of heat absorbed from the outside air is
Explanation of Solution
Write the expression for the rate of conversation of energy principle for refrigerant 134a.
Here, the rate of heat rejected in the condenser is
Conclusion:
Substitute
Thus, the rate of heat absorbed from the outside air is
Want to see more full solutions like this?
Chapter 6 Solutions
Thermodynamics: An Engineering Approach
- How does the efficiency of the Stirling engine compare to the Carnot engine working within the same two heat reservoirs?arrow_forwardRefrigerant-134a is to be cooled by water in a condenser. The refrigerant enters the condenser with a mass flow rate of 6 kg/min at 1 MPa and 70°C and leaves at 35°C. The cooling water enters at 300 kPa and 15°C and leaves at 25°C. Neglecting any pressure drops, determine the heat transfer rate from the refrigerant to water.arrow_forwardRefrigerant-134a is to be cooled by water in a condenser. The refrigerant enters the condenser with a mass flow rate of 6 kg/min at 1 MPa and 70°C and leaves at 35°C. The cooling water enters at 300 kPa and 15°C and leaves at 25°C. Neglecting any pressure drops, determine the mass flow rate of the cooling water required.arrow_forward
- Refrigerant-134a enters the evaporator coils placed at the back of the freezer section of a household refrigerator at 100 kPa with a quality of 20 percent and leaves at 100 kPa and −26°C. If the compressor consumes 600 W of power and the COP of the refrigerator is 1.2, determine the rate of heat rejected to the kitchen air.arrow_forwardConsider an adiabatic turbine operating steadily. Does the work output of the turbine have to be equal to the decrease in the energy of the steam flowing through it?arrow_forwardRefrigerant-134a at a rate of 0.08 kg/s enters the compressor of a refrigerator as superheated vapor at 0.18 MPa and 0 ℃ and leaves at 0.9 MPa and 80 ℃. The refrigerant is cooled in the condenser to 31.3 ℃ and 0.8 MPa and it is throttled to 0.18 MPa. Disregarding any heat transfer and pressure drops in the connecting lines between the components, Determine the coefficient of performance of the refrigerator.arrow_forward
- At a rate of 0.05 kg/s, refrigerant-134a enters the compressor of a refrigerator as superheated vapour at 0.14 MPa and -10 oC, and exits at 0.8 MPa and 50 oC. The refrigerant is cooled to 26°C and 0.72 MPa in the condenser before being throttled to 0.15 MPa. Determine: Taking into account any heat transfer and pressure drops in the connecting lines between the components, a. The isentropic efficiency of the compressor b.The coefficient of performance of the refrigeratorarrow_forwardSteam enters an adiabatic turbine with a temperature of 300°C and a pressure of 4MPa, then leaves with a temperature of 200°C and a pressure of 1MPa. Calculate the isentropic efficiency of the turbine and determine if it is in its normal range.arrow_forwardDoes a refrigerator that has a higher COP necessarily have a higher second-law efficiency than one with a lower COP? Explain.arrow_forward
- Refrigerant-134a at 1600 kPa and 80°C is expanded adiabatically in a closed system to 100 kPa with an isentropic expansion efficiency of 85 percent. Determine the second-law efficiency of this expansion. Take T0 = 25°C and P0 = 100 kPa.arrow_forwardRefrigerant 134-a enters the coil of the condenser of a residential heat pump at 500kPa and 400C and leaves at the same pressure as a saturated liquid at a rate of 0.05kg/s. Determine the power of a compressor required to run the heat pump if heat absorption from the outside air is 200kJ/kg. Please give clear solution and step by steparrow_forwardWhy is it important to clean the condenser coils of a household refrigerator a few times a year? Also, why is it important not to block airflow through the condenser coils?arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





