
Essentials of Biology (5th International Edition)
5th Edition
ISBN: 9781259660269
Author: Sylvia S. Mader, Dr., Michael Windelspecht
Publisher: Mcgraw-Hill
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.2, Problem 3A
When electrons in the reaction center of PS 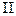 are passed to an energy-acceptor molecule, they are replaced by electrons that have been given up by
are passed to an energy-acceptor molecule, they are replaced by electrons that have been given up by
a. oxygen.
b. glucose.
c. carbon dioxide.
d. water.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Substance A is converted to substance B in a metabolic reaction. Which statement best describes the role of enzyme during this reaction?
A. It dissolves substance A in the reaction medium.
B. It speeds up the reaction without it being consumed in the process.
C. It provides energy to the reaction system
D. It adjusts the pH of the reaction medium.
Enzymes catalyze a chemical reaction by
A. decreasing the amount of ATP required to activate a reaction
B. increasing the amount of ATP required to activate a reaction
C. slowing down the reaction to obtain more products
D. increasing the reaction to obtain more products
E. decreasing the amount of ATP required and obtaining more products.
Which of the following is not true about enzymes: a. They increase ΔG of reactions. b. They are usually made of amino acids. c. They lower the activation energy of chemical reactions. d. Each one is specific to the particular substrate(s) to which it binds.
Chapter 6 Solutions
Essentials of Biology (5th International Edition)
Ch. 6.1 - Describe the function of the chloroplast in...Ch. 6.1 - Prob. 2LOCh. 6.1 - Prob. 3LOCh. 6.1 - Prob. 1CYPCh. 6.1 - Prob. 2CYPCh. 6.1 - Distinguish between a reduction and an oxidation...Ch. 6.1 - The raw materials for photosynthesis are a. oxygen...Ch. 6.1 - Prob. 2ACh. 6.2 - Prob. 1LOCh. 6.2 - Prob. 2LO
Ch. 6.2 - Prob. 3LOCh. 6.2 - Prob. 1CYPCh. 6.2 - Prob. 2CYPCh. 6.2 - Describe the path of electrons from water to NADPHCh. 6.2 - Prob. 4CYPCh. 6.2 - When electrons in the reaction center of PS are...Ch. 6.2 - Prob. 4ACh. 6.2 - Prob. 5ACh. 6.2 - Prob. 6ACh. 6.3 - Prob. 1LOCh. 6.3 - Prob. 2LOCh. 6.3 - Prob. 3LOCh. 6.3 - Prob. 1CYPCh. 6.3 - Prob. 2CYPCh. 6.3 - Prob. 3CYPCh. 6.3 - Prob. 7ACh. 6.3 - Prob. 8ACh. 6.3 - The output of the Calvin cycle that is used to...Ch. 6.4 - Prob. 1LOCh. 6.4 - Prob. 2LOCh. 6.4 - Prob. 3LOCh. 6.4 - Prob. 4LOCh. 6.4 - Identify the differences between C 3 and C 4...Ch. 6.4 - Prob. 2CYPCh. 6.4 - Prob. 3CYPCh. 6.4 - Prob. 10ACh. 6.4 - Prob. 11ACh. 6 - Prob. S1.1BYBCh. 6 - Prob. S4.4BYBCh. 6 - Prob. S5.2BYBCh. 6 - Prob. 1TCCh. 6 - Prob. 2TCCh. 6 - Prob. 3TCCh. 6 - Prob. 4TCCh. 6 - Prob. 5TC
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- When an enzymatic reaction is in progress, do you expect to see happen to the amount of substrate? A It will increase as the reaction proceeds. B It will decrease as the reaction proceeds. C The amount will not change as reaction proceeds. D It is not possible to predict what will happen to it as the reaction proceeds.arrow_forwardWhat is a regulatory mechanism in which a reaction product slows or stops a metabolic pathway that produces it? A. Phosphorylation B. Feedback inhibition C. Electron transfer chain D. Coenzymearrow_forwardWhich of the following statement regarding enzymes is false? a.) they speed up reaction rates b.) they work within narrow temperature and pH ranges c.) The active site binds the substarate d.) They are consumed in the reactions they catalyzearrow_forward
- When an enzymatic reaction is in progress, do you expect to see happen to the amount of product? A It will increase as the reaction proceeds. B It will decrease as the reaction proceeds. C The amount will not change as reaction proceeds. D It is not possible to predict what will happen to it as the reaction proceeds.arrow_forwardWhich of these statements about enzymes is true? a,Most proteins are enzymes. b.Most enzymes are proteins. c.Enzymes are changed by the reactions they catalyze. d.The active sites of enzymes have little specificity for substrates.arrow_forwardWhich of the following accurately describes how an enzyme functions? a. reduces the energy content of the products b. allows for the exothermic reaction of materials that are usually endothermic c. changes the reaction mechanism effectively reducing the activation energy d. shifts the equilibrium for the reactionarrow_forward
- Which of the following is NOT true of enzymes? a) They may interact chemically with their substrates b) They may catalyze a reaction in both forward and reverse directions. c) They may alter Keq for the reaction that they catalyze. d) They may decrease the entropy of their substrates.arrow_forwardenzymes are efficient catalysts because they can do which of the following? A) catalyze reactions that ottherwise would not occur. B) decrease the free energy of activation of reactions, C) decrease the standard free energy change o reactions , D) prevent the conversion of product to substrate, E) shift the equilibrium of reactions toward more complete inversion of productarrow_forwardWhich of the following is correct regarding ATP?a. It is an amino acid.b. It has a helical structure.c. It is a high-energy molecule that can break down to ADP andphosphate.d. It is a nucleotide component of DNA and RNA.arrow_forward
- Which statement is true regarding the function of a catalyst in a chemical reaction? a) A catalyst increases the rate of a reaction. b) A catalyst provides an alternate mechanism for the reaction. c) A catalyst is not consumed by the reaction. d) All of the above are true.arrow_forwardIn a highly high-temperature environment, you placed an enzyme and some substrates. You've seen that there hasn't been any reaction. Applying what you have learned on enzyme activity, how would you explain what occurred? A. The enzyme was not placed at a pH level that would allow it to react at its maximum rate. B. There are not enough enzymes for the reaction to occur. C. The enzyme may have denatured or broken down due to the high temperature of the environment, resulting in no reaction. D. There are not enough substrates for reaction to occur. Substrate concentration affects enzyme activity. Determine what would occur when there are too many substrates and fewer enzymes? A. No enzyme activity would occur. B. The reaction rate would continuously increase. C. The reaction would speed up tremendously. D. Maximal rate of reaction would occur. Enzyme activity mainly involves the active site and the substrate. How would you compare the active site from the substrate? A. The substrate…arrow_forwardWhich of the following is TRUE about the enzymatic catalysis of a chemical reaction? A. It increases the energy of the transition state B. It decreases the free energy ∆G so that a reaction can proceed spontaneously C. It decreases the energy of the transition state D. It increases the forward and reverse ratesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College
Theory of Spontaneous generation | Abiogenesis and Biogenesis |; Author: subrata das;https://www.youtube.com/watch?v=tcyESFngVPk;License: Standard YouTube License, CC-BY