
Concept explainers
(a)
Interpretation:
For the given set of species the available
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Leaving group: it is a fragment that leaves substrate with a pair of electrons via heterolytic bond cleavage.
Cation: The positively charged chemical species is referred as cation.
(b)
Interpretation:
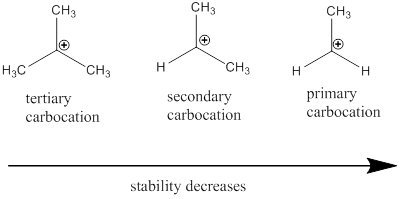
From the given carbocations the most stable one should be identified.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Cation: The positively charged chemical species is referred as cation.
Carbocation stability order:
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry
- 9. Which of the following alkenes has a Z configuration?arrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energy. (continued)762 CHAPTER 15 Conjugated Systems, Orbital Symmetry, and Ultraviolet Spectroscopy(e) Show how many electrons are in each MO for the pentadienyl radical (ground state).(f) Show how your molecular orbital picture agrees with the resonance picture showing delocalization of the unpairedelectron onto three carbon atoms.(g) Remove the highest-energy electron from the pentadienyl radical to give the pentadienyl cation. Which carbon atomsshare the positive charge? Does this picture agree with the resonance picture?(h) Add an…arrow_forwardThe pentadienyl radical, H2C“CH¬CH“CH¬CH2#, has its unpaired electron delocalized over three carbon atoms.(a) Use resonance forms to show which three carbon atoms bear the unpaired electron.(b) How many MOs are there in the molecular orbital picture of the pentadienyl radical?(c) How many nodes are there in the lowest-energy MO of the pentadienyl system? How many in the highest-energy MO?(d) Draw the MOs of the pentadienyl system in order of increasing energyarrow_forward
- Are phenyl carbocations more stable than tertiary carbocations? Is there a carbocation that is more stable than a tertiary carbocation? If yes, what is the reason why it is more stablearrow_forwardHow would you develop a molecular orbital for a structure like buta-2-diene that has an anion on the first carbon and a cation on the fourth carbon? Also, how would MO look like for 2-dimethyl-1,3-pentadiene?arrow_forwardBased on reaction 1, how many isomers can be formed? Is the isomer cis or trans? The target molecule and reaction 1 are included.arrow_forward
- The central carbon atom of an allene is a member of two double bonds, and ithas an interesting orbital arrangement that holds the two ends of the molecule at rightangles to each other.(a) Draw an orbital diagram of allene, showing why the two ends are perpendicular.(b) Draw the two enantiomers of penta-2,3-diene. A model may be helpfularrow_forwarda. Write a second resonance structure for the following carbocation. b. Is the charge shared equally by both allylic carbons? If not, which one bears more of the charge? Select the single best answer. Both carbocations carry the same amount of charge. The structure on the left in Part 1 contributes more to the resonance hybrid. The structure on the right in Part 1 contributes more to the resonance hybrid.arrow_forwarddraw the two possible carbocations that can form when this alkene reacts with a strong acid (such as HBr or H3O+). of the two structures you drew, circle the more stable carbocationarrow_forward
- Answer the following questions for the molecular orbitals (MOs) of 1,3,5,7-octatetraene: a. How many MOs does the compound have? b. Which are the bonding MOs, and which are the antibonding MOs? c. Which MOs are symmetric, and which are antisymmetric? d. Which MO is the HOMO and which is the LUMO in the ground state? e. Which MO is the HOMO and which is the LUMO in the excited state? f. What is the relationship between HOMO and LUMO and symmetric and antisymmetric orbitals? g. How many nodes does the highest-energy MO of 1,3,5,7-octatetraene have between the nuclei?arrow_forwardPlease draw the line structures of all of the C8H17+ carbocations that have the carbon-skeleton below. And circle the most stable carbocation of those drawn.arrow_forwardClassify attached carbocation as 1°, 2°, or 3°.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


