
ORGANIC CHEMISTRY-OWL V2 ACCESS
8th Edition
ISBN: 9781305582422
Author: Brown
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6.3, Problem 6.3P
Interpretation Introduction
Interpretation:
The carbocations should be arranged in increasing stability order.
Concept Introduction:
Carbocation: The carbon ion that bears a positive charge on it is termed as carbocation.
Carbocation stability order:
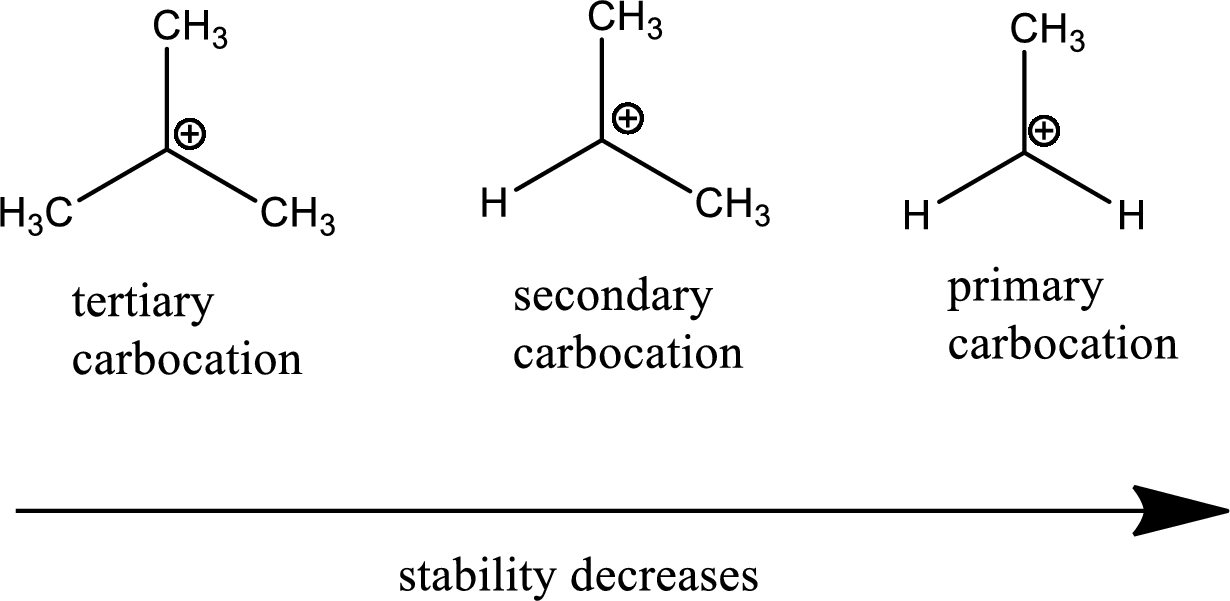
The tertiary carbocation is more stable than secondary and primary since tertiary carbocation contains 3 methyl groups (electron releasing) in which the positive charge is distributed. The more positive charge gets distributed, more stable the compound. Therefore, the tertiary is more stable than secondary and secondary is more stable than primary.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Derive an IUPAC name for the following (cyclo)alkenes.
(Do not use cis/trans in your names. Use only the (E)/(Z) designations for double bond stereochemistry. It is not necessary to use italics in writing compound names.)
(a)
(b)
my
X
11:43
Q1.
(a)
(c)
(d)
(b) Two stereoisomers of but-2-ene are formed when 2-bromobutane reacts with
ethanolic potassium hydroxide.
(i) Explain what is meant by the term stereoisomers.
Library
Name and outline a mechanism for the reaction of 2-bromo-2-methylpropane with
ethanolic potassium hydroxide to form the alkene 2-methylpropene, (CH3)2C=CH₂
Name of mechanism
Mechanism
(ii)
Draw the structures and give the names of the two stereoisomers of
but-2-ene.
Stereoisomer 1
Name
(iii) Name this type of stereoisomerism.
Select
Name
Stereoisomer 2
When 2-bromo-2-methylpropane reacts with aqueous potassium hydroxide,
2-methylpropan-2-ol is formed as shown by the following equation.
CH3
H₂C-C-CH3 + KOH
Br
Page 2 of 14
CH3
H3C-C-CH3 + KBr
ОН
State the role of the hydroxide ions in this reaction.
Write an equation for the reaction that occurs when CH3CH₂CH₂CH₂Br reacts with
an excess of ammonia. Name the organic product of this reaction.
Equation
Name of product
9,284 Photos, 1,166 Videos
For You…
For each molecular formula, draw all the isomeric alkynes, and give their IUPAC names.Circle the acetylenic hydrogen of each terminal alkyne.(a) C5H8 (three isomers) (b) C6H10 (seven isomers)
Chapter 6 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
Ch. 6.2 - Using the BDE values from Appendix 3, calculate...Ch. 6.3 - Name and draw a structural formula for the product...Ch. 6.3 - Prob. 6.3PCh. 6.3 - Propose a mechanism for the addition of HI to...Ch. 6.3 - Prob. 6.5PCh. 6.3 - Propose a mechanism for the acid-catalyzed...Ch. 6.3 - The acid-catalyzed hydration of...Ch. 6.3 - Complete these reactions. (a) (b)Ch. 6.3 - Draw the structure of the chlorohydrin formed by...Ch. 6.4 - Draw structural formulas for the alkene that gives...
Ch. 6.5 - Prob. 6.11PCh. 6.5 - Prob. 6.12PCh. 6.5 - What alkene with the molecular formula C6H12, when...Ch. 6 - Prob. 6.15PCh. 6 - Prob. 6.16PCh. 6 - Predict the organic product(s) of the reaction of...Ch. 6 - Prob. 6.18PCh. 6 - Prob. 6.20PCh. 6 - Draw a structural formula for an alkene with the...Ch. 6 - Account for the fact that addition of HCl to...Ch. 6 - Account for the fact that treating propenoic acid...Ch. 6 - Draw a structural formula for the alkene with the...Ch. 6 - Draw the alternative chair conformations for the...Ch. 6 - Draw a structural formula for the cycloalkene with...Ch. 6 - Reaction of this bicycloalkene with bromine in...Ch. 6 - Terpin, prepared commercially by the...Ch. 6 - Propose a mechanism for this reaction and account...Ch. 6 - Treating 2-methylpropene with methanol in the...Ch. 6 - When 2-pentene is treated with Cl2 in methanol,...Ch. 6 - Treating cyclohexene with HBr in the presence of...Ch. 6 - Propose a mechanism for this reaction. 1-Pentane...Ch. 6 - Treating 4-penten-1-ol with bromine in water forms...Ch. 6 - Prob. 6.35PCh. 6 - Prob. 6.36PCh. 6 - Reaction of -pinene with borane followed by...Ch. 6 - Write structural formulas for the major organic...Ch. 6 - Draw the structural formula of the alkene that...Ch. 6 - Consider the following reaction. (a) Draw a...Ch. 6 - Prob. 6.42PCh. 6 - Prob. 6.43PCh. 6 - Show how to convert ethylene to these compounds....Ch. 6 - Show how to convert cyclopentene into these...Ch. 6 - Prob. 6.46PCh. 6 - Describe the stereochemistry of the bromohydrin...Ch. 6 - Prob. 6.49PCh. 6 - Treating 1,3-butadiene with 1 mole of HBr gives a...Ch. 6 - In this chapter, we studied the mechanism of the...Ch. 6 - As we have seen in this chapter, carbon-carbon...Ch. 6 - Prob. 6.53PCh. 6 - Prob. 6.54P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the given carbocation in orbital phase. CH2 CH2 (b) (a) (d)arrow_forwardProvide the IUPAC name for each of the following compounds. Pay close attention to stereochemistry. (a) (b) -NH2 (c) OH ОН NH2 NH2 НО .CI Br. NH2 O,N"arrow_forward1. (a) Describe aromaticity, Kekule structure and resonance structure for benzene. (b) Why is benzene more stable than aliphatic alkenes?arrow_forward
- a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomeric products are formed. Give their structures, and label each asymmetric carbon atom as (R) or (S). What is the relationship between these isomers?arrow_forwardGive IUPAC names for the following structures. (If appropriate, specify relative stereochemistry.) (a) (b) S Sarrow_forwardUsing cyclohexane as your starting material, show how you would synthesize each of the following compounds. (Onceyou have shown how to synthesize a compound, you may use it as the starting material in any later parts of this problem.)(a) bromocyclohexane (b) cyclohexenearrow_forwardDisiamylborane adds only once to alkynes by virtue of its two bulky secondary isoamylgroups. Disiamylborane is prepared by the reaction of BH3 # THF with an alkene.(a) Draw the structural formulas of the reagents and the products in the preparation ofdisiamylborane.(b) Explain why the reaction in part (a) goes only as far as the dialkylborane. Why isSia3B not formed?arrow_forward(a) Draw the products formed when propane is heated with Br2. (b) Label major and minor products and explain with an energy diagram.arrow_forwardFor each molecular formula, draw all the isomeric alkynes, and give their IUPAC names.Circle the acetylenic hydrogen of each terminal alkyne.(a) C5H8 (three isomers)arrow_forward1 Provide the IUPAC name of the following compounds, with clear indication of stereochemistry for stereocenters and alkene. (a) (b) Br (c) Me. (d) Mearrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY