
Interpretation:
The pathway for solving a “grams of A” to “grams of B” by the use of conversion factor has to be chosen from the given options.
Concept Introduction:
Using the

Molar mass of a substance gives a relationship between the number of grams and number of moles of that particular substance.

The Chemical formula subscript also provides relationship between the number of moles of substance to the number of moles of its individual components present in the substance.

All the three above relationship can be put into a single diagram as shown below,
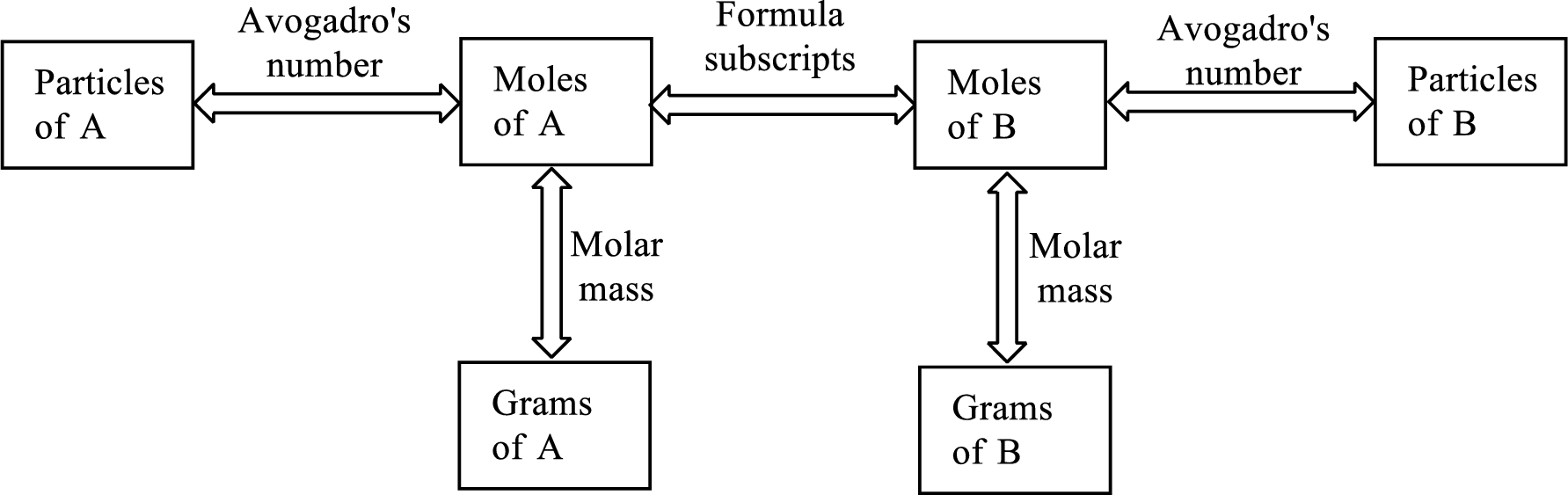
Trending nowThis is a popular solution!

Chapter 6 Solutions
General, Organic, and Biological Chemistry
- The problem How many grams of O2 are needed to produce 2.00 moles of H2O via the chemical reaction 2H2 + O2 2H2O? is characterized as a a. moles of A to grams of A problem b. grams of A to moles of B problem c. moles of A to grams of B problem d. no correct responsearrow_forwardInitial Temp of HCl: 22.96 C Final Temp of HCl: 28.30 C Moles of HCl: .05 moles Moles of NaOH: .05 moles Cs water: 4.184 J/g C Accepted delta Hrxn: -55.8 kJ/mol 1. what is the qwater. pls include formula. 2. calculate delta Hrxnn using the moles, temp and cswater 3. calculate delta Hrxn % errorarrow_forwardGive answer to all parts? Initial Temp of HCl: 22.96 C Final Temp of HCl: 28.30 C Moles of HCl: .05 moles Moles of NaOH: .05 moles Cs water: 4.184 J/g C Accepted delta Hrxn: -55.8 kJ/mol 1. what is the qwater. pls include formula. 2. calculate delta Hrxnn using the moles, temp and cswater 3. calculate delta Hrxn % errorarrow_forward
- A fertilizer has the analysis of 12-12-12. How many kilograms of N is there in the 300 kg of fertilizer? Numeric answer only.arrow_forwardAnswer 1 and 2 PROBLEM SOLVING. Show all pertinent computations Express final answer up to the fourth decimal place. 1.ln the preparation of aspirin, 6.0 grams of salicylic acid was made to react with acetic anhydride gnbxdride forming 4 grams of aspirin. Calculate the % yield of aspirin. 2. calculate the amount of sodium salt of aspirin obtained from the preparation.arrow_forward____C3H8 + _5_O2 ➞ _3_CO2 + _4_H2O Reaction Type:______ How many moles of oxygen are required to react with 12.5 g C3H8? show work in answerarrow_forward
- 4 C3H6 (g) + 6 NO (g) → 4 C3H3N (g) + 6 H2O (g) + N2 (g)• How many grams of NO would be left over (unreacted) when 30.00 g of C3H6 reacts with 55.00 g of NO?Answer grams of NOMolecular WeightsC3H6 42.081 g/molNO 30.006 g/molC3H3N 53.064 g/molH2O 18.015 g/molN2 28.014 g/molarrow_forwardHow many moles of NO2 you should obtain if 0.60 mol O2 reacts with excess of NO? 2 NO + O2 ------> 2 NO2 Round result to two places past decimalarrow_forwardWhat is the correct form of the conversion factor needed to convert the number of moles of H2O to the number of moles of Mg(OH)2 produced? Mg3N2 + 6H2O → 3Mg(OH)2 + 2NH3 Answer Choices: 1/6 2/6 6/2 18/17 1/2arrow_forward
- Instruction: Final answer should be expressed with least number of significant figures. Question: 100.0 grams of sodium sulfate reacts with 50.00 grams of barium nitrate. A-C IS ALREADY ANSWERED. ANSWER D-F QUESTIONS. a) What is the balanced equation? b) How many grams of barium sulfate will form? c) What is the limiting reactant? d) How much limiting reactant was used up in the reaction? e) How much excess reactant was used up for this reaction? f) How much of the excess reactant was left?arrow_forwardN2 + 3 H2 ==> 2 NH3 How many grams of ammonia is produced if we react 11.0 grams of nitrogen Your Answer:arrow_forward4fe+3o2⟶2fe2o3 how many grams of Fe2O3 will be produced from 37.5 grams of iron? show work.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
