
Concept explainers
Using the equilibrium mixture of reactants and products, calculate K for the reaction with the balanced equation,
More Practice: Try Problems 6.51, 6.52, 6.59, 6.60.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
Additional Science Textbook Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Organic Chemistry
Thermodynamics, Statistical Thermodynamics, & Kinetics
Principles of General, Organic, Biological Chemistry
General, Organic, & Biological Chemistry
- lum constant Suppose a 500. mL flask is filled with 0.30 mol of I, and 1.9 mol of HI. The following reaction becomes possible: H,(g) + I,(g) = 2 HI(g) The equilibrium constant K for this reaction is 5.08 at the temperature of the flask. Calculate the equilibrium molarity of I,. Round your answer to two decimal places. M Check 2021 McG N Explgationarrow_forwardWhich of the following statements is (are) true? (Select all that apply.) If equilibrium constants for the chemical reaction N2(g) + O2(g) : = 2 NO(g) are K, = 1.1 x 103 and 3.6 x 103 at 2,200 K and 2,500 K, respectively, then the reaction is exothermic. If Q < K then the reactant concentrations will be decrease as the reaction proceeds to equilibrium. Concentration of the products equals the concentrations of the reactants. If the reaction, C(s) + 2H2(g) = CH4(g) is at equilibrium, adding solid carbon will cause the reaction to shift in the forward direction. None of these statements are true. O At equilibrium, the rate of the forward reaction is equal to the rate of the reverse reaction.arrow_forwardSuppose a 500. mL flask is filled with 0.90 mol of NO, and 0.20 mol of NO,. The following reaction becomes possible: NO, (g) + NO(g) 2NO,(8) The equilibrium constant K for this reaction is 0.556 at the temperature of the flask. Calculate the equilibrium molarity of NO. Round your answer to two decimal places. Explanation Check Terma of Use 2021 McGraw-Hill Education. All Rights Reservedarrow_forward
- At 448°C, the equilibrium constant for the reaction: H₂+I22 HI is 50.5. What concentration of I2 would be found in an equilibrium mixture in which the concentrations of HI and H₂ were 0.540 M and 0.055 M, respectively? Submit Answer M Try Another Version 10 item attempts remainingarrow_forwardtemperature. Practice Exercise At 1280°C the equilibrium constant (K) for the reaction Br2(g) 2Br(g) is 1,1 x 10, If the initial concentrations are [Br2] = 6.3 X 102 M and [Br] = 1.2 x 10 M, calculate the concentrations of these species at equilibrium. %3Darrow_forwardSuppose a 250. mL flask is filled with 0.30 mol of H, and 1.3 mol of HCI. The following reaction becomes possible: H, (2) + Cl, (8) - 2HCI(g) The equilibrium constant K for this reaction is 5.49 at the temperature of the flask. Calculate the equilibrium molarity of H,. Round your answer to two decimal places. IM Check Explanation 2021 McGraw-Hill Education. All Rights Reserved. acerarrow_forward
- 3 Question 40. Listen For the reaction given below, the value of the equilibrium constant at 400 K is 64.0. Br₂(g) + Cl₂(g) Math output error 2BrCl(g) At equilibrium, the concentrations of Br2 and Cl2 are each 0.18 mol/L. What is the equilibrium concentration of BrCl in M? Your Answer Answer unitsarrow_forwardCHEMWORK The reaction H,(g) + I(g) 2 2HI(g) has K, = 45.9 at 763 K. A particular equilibrium mixture at 763 K contains HI at a pressure of 3.28 atm and H, at a pressure of 1.20 atm. Calculate the equilibrium pressure of I,(g) in this mixture. atm PL Submit Submit Answer Try Another Version 1 item attempt remaining (Previous ywww.yahoo.com 2h A Top Story New research raises doubts about 1 st COVID-19 death count Cengage Learning | Cengage Technical Support DELLarrow_forwardThe equilibrium constant for the reaction NH3 (g) + HF (g) at 25 °C is 10.2. ? NH4F (S) (a) If the equilibrium partial pressure of ammonia is P °C? Submit Answer = 0.926 atm and solid ammonium fluoride is present, what is the equilibrium partial pressure of hydrogen fluoride at 25 NH3 Try Another Version P = HF (b) An excess of solid NH4F is added to a container filled with NH3 at 25 °C and a pressure of 1.08 atm. Calculate the pressures of NH3 (g) and HF (g) reached at equilibrium. (For each answer, enter an unrounded value.) P P 5 item attempts remaining NH3 = HF atm = atm atmarrow_forward
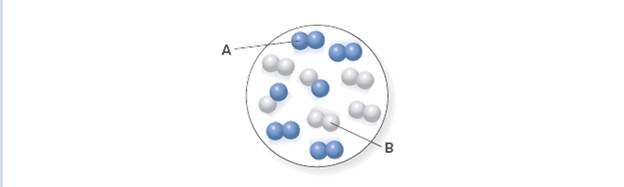
- The following figure represents an equilibrium mixture of reactant A (red spheres) and product B (blue). The associated chemical reaction is A(g) = 2B(g). Assume the volume of the box is 1.0 L and that each sphere represents 1 mole of A or B. 1st attempt See Periodic Table What is the value of the equilibrium constant K?arrow_forwardChapter 10 Homework Problem 10.49 10 of 17 Review I Constants I Periodic Table Consider the reaction: Part B 2NH3 (g) =N2(g) + 3H2(g) What is the numerical value of K. for the reaction if the concentrations at equilibrium are 0.16 M NH3,5.0 M N2, and 0.60 M H2 ? Express your answer using two significant figures. Kc = Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining « Previous Next 80 888arrow_forwardFor the reaction PCI, (g) + Cl,(g) = - PCI, (g) the initial mixture contains 4.85 mol PCI3, 5.83 mol Cl2, and no moles of PCI5. At equilibrium, 1.30 mol of PCl3remains. 1st attempt See Periodic Table See Hint How many moles of Cl2 and PCI5 are present at equilibrium? (Be sure to give your answer to two decimal places.) mol Cl2 mol PCI5arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





