
ORGANIC CHEMISTRY 2-SEMESTER-ACCESS
4th Edition
ISBN: 9781119659532
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6.7, Problem 10ATS
Interpretation Introduction
Interpretation: The resonating structures of R-(-)-carvone must be interpreted to show all the electrophilic centers.
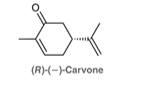
Concept introduction: An electrophile is an electron-deficient species whereas a nucleophile is an electron-rich species. During a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Under certain reaction conditions, 2,3-dibromobutane reacts with two equivalents of base to give three products, each of which contains two new p bonds. Product A has two sp hybridized carbon atoms, product B has one sp hybridized carbon atom, and product C has none. What are the structures of A, B, and C?
Reaction of (CH3)3CH with Cl2 forms two products: (CH3)2CHCH2Cl (63%) and (CH3)3CCl (37%). Why is the major product formed by cleavage of the stronger 1° C–H bond?
CH3
H
НС— С—С—С—ОН
H
NH,
Valine is one of the 20 amino acids used to form proteins, it contains both a carboxylic acid group and an amino group. Draw the structure of valine as it would appear at pH 7.
• You do not have to consider stereochemistry.
P.
opy aste
Chapter 6 Solutions
ORGANIC CHEMISTRY 2-SEMESTER-ACCESS
Ch. 6.1 - Prob. 1LTSCh. 6.1 - Prob. 1PTSCh. 6.1 - Prob. 2ATSCh. 6.2 - Prob. 3CCCh. 6.3 - Prob. 4CCCh. 6.3 - Prob. 5CCCh. 6.4 - Prob. 6CCCh. 6.6 - Prob. 7CCCh. 6.7 - Prob. 2LTSCh. 6.7 - Prob. 8PTS
Ch. 6.7 - Prob. 9PTSCh. 6.7 - Prob. 10ATSCh. 6.8 - Prob. 3LTSCh. 6.8 - Prob. 11PTSCh. 6.8 - Prob. 12ATSCh. 6.9 - Prob. 4LTSCh. 6.9 - Prob. 13PTSCh. 6.9 - Prob. 14ATSCh. 6.10 - Prob. 5LTSCh. 6.10 - Prob. 15PTSCh. 6.10 - Prob. 16ATSCh. 6.11 - Prob. 6LTSCh. 6.11 - Prob. 17PTSCh. 6.11 - Prob. 18ATSCh. 6 - Prob. 19PPCh. 6 - Prob. 20PPCh. 6 - Prob. 21PPCh. 6 - Prob. 22PPCh. 6 - Prob. 24PPCh. 6 - Prob. 25PPCh. 6 - Prob. 26PPCh. 6 - Prob. 27PPCh. 6 - Prob. 28PPCh. 6 - Prob. 29PPCh. 6 - Prob. 30PPCh. 6 - Prob. 31PPCh. 6 - Prob. 32PPCh. 6 - Prob. 33PPCh. 6 - Prob. 34PPCh. 6 - Prob. 35PPCh. 6 - Prob. 36PPCh. 6 - Prob. 37PPCh. 6 - Prob. 38PPCh. 6 - Prob. 39PPCh. 6 - Prob. 40PPCh. 6 - Prob. 41PPCh. 6 - Prob. 43ASPCh. 6 - Prob. 44ASPCh. 6 - Prob. 45ASPCh. 6 - Prob. 46ASPCh. 6 - Prob. 47ASPCh. 6 - Prob. 48ASPCh. 6 - Prob. 49ASPCh. 6 - Prob. 50IPCh. 6 - Prob. 51IPCh. 6 - Prob. 52IPCh. 6 - Prob. 53IPCh. 6 - Prob. 54IPCh. 6 - Prob. 55IPCh. 6 - Prob. 56IPCh. 6 - Prob. 57IPCh. 6 - Prob. 58IPCh. 6 - Prob. 59IPCh. 6 - Prob. 60IPCh. 6 - Prob. 61IPCh. 6 - Prob. 62CPCh. 6 - Prob. 64CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw a resonance structure, complete with all formal charges and lone (unshared) electron pairs, that shows the resonance interaction of the acetoxy with the para position in phenyl acetate. phenyl acetate ● CH3 • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. In cases where there is more than one answer, just draw one. Sn [F ?arrow_forwardCyclic forms of D-glucose: Furanose and Pyranose Recall that aldehyde and alcohol react to form hemiacetal. The O in OH acts as nucleophile, attacking the partially positive C of C=O. The H from the OH of alcohol is transferred to the O of C=O. он R R1 HO, On the other hand, ketone and alcohol also react to form hemiketal. OH R R2 R R1 HO, In glucose the OH in C-4 or C-5 can react with the C=O. If the C-4 OH reacts with the C=O the structure is a furanose (5-membered ring); if C-5 OH the product is a pyranose (6-membered ring). Furanose and pyranose are derived from cyclic ethers furan and pyran. Both furanose and pyranose have two isomers: the alpha and beta (also called anomers). Furan Pyran Furanose and pyranose form of D-glucose (note the numbering of carbon atoms) Furanose (5-membered ring) Alpha Pyranose (6-membered ring) Alpha Beta Beta 1 HO- Но H- Но H- _2 FH- H- OH FHO- H HO. но- H- но- H. Но Но H-4 H_5. FHO- FHO- H OH 6CH2OH H CH,OH CH,OH CH2OH C-1 OH is on the right Alpha-D-…arrow_forwardN,N-diethyl-m-toluamide (DEET) is the active ingredient in many insect repellent preparations. Following is one of the steps in its synthesis. In the box below draw the structure of the product of this reaction. H3C MgBr 1. CO2 2. H3O+ product • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw the Grignard reagent as a covalent magnesium bromide. 90-87 0 + 11 ? n [arrow_forward
- 2,4,6-Trinitrophenol is known by the common name picricacid. Picric acid is a solid but is readily soluble in water. Insolution, it is used as a biological tissue stain. As a solid,it is also known to be unstable and may explode. In thisway, it is similar to 2,4,6-trinitrotoluene (TNT). Draw thestructures of picric acid and TNT. Why is picric acid readily soluble in water, whereas TNT is not?arrow_forwardIn an advanced synthetic chemistry experiment, a researcher prepares a compound, ZY-7, by reacting a ketone (C5H10O) with hydroxylamine (NH2OH), followed by heating in the presence of an acid catalyst. The resulting compound, ZY-7, is then treated with a solution of sodium nitrite (NaNO2) and hydrochloric acid (HCl) at low temperature. Identify the class of compound that ZY-7 most likely belongs to after this series of reactions." A) Amide B) Oxime C) Nitro compound D) Diazonium salt E) Ester Don't use chatgpt please provide valuable answerarrow_forwardExplain why triphenylene resembles benzene in that it does not undergo addition reactions with Br2, but phenanthrene reacts with Br2 to yield the addition product drawn. (Hint: Draw resonance structures for both triphenylene and phenanthrene, and use them to determine how delocalized each π bond is.)arrow_forward
- Briefly, but clearly, explain why the –OH hydrogen in acetic acid (CH3CO2H) is more acidic than in ethanol (C2H5OH).arrow_forward4-pyranone will readily undergo an acid-base reaction. Identify the reaction conditions that will result in the formation of an aromatic product. Then, draw the aromatic resonance product structure. Include all lone pairs in your structure. Ignore inorganic byproducts. H3O+arrow_forwardDraw a structural formula for the substitution product of the reaction shown below. H/ ● (CH3)3C |YY Br ΔΟ H Na • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • If more than one stereoisomer of product is formed, draw both. Separate multiple products using the + sign from the drop-down menu. • Products that are initially formed as ions should be drawn in their neutral forms. SH acetone [Farrow_forward
- CH=CHCN 3-phenylpropenenitrile Electrophilic substitution on 3-phenylpropenenitrile occurs at the meta position. Draw resonance structures to show how the ring is electron-poor at the ortho and para positions. ● • You do not have to consider stereochemistry. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate resonance structures using the → symbol from the drop-down menu.arrow_forwardIn an advanced synthetic chemistry experiment, a researcher prepares a compound, ZY-7, by reacting a ketone (C5H100) with hydroxylamine (NH2OH), followed by heating in the presence of an acid catalyst. The resulting compound, ZY-7, is then treated with a solution of sodium nitrite (NaNO2) and hydrochloric acid (HCI) at low temperature. Identify the class of compound that ZY-7 most likely belongs to after this series of reactions." A) Amide B) Oxime C) Nitro compound D) Diazonium salt E) Ester Don't use chatgpt please provide valuable answerarrow_forwardEt, NH no reaction However, when the carbon-carbon double bond has a carbonyl group adjacent to it, the double bond reacts readily with nucleophiles by nucleophilic addition. nucleophilic addition Et NH + For the following reaction, draw the structure of the resonance contributor that is attacked by diethylamine. Et, NH + • Include all valence lone pairs in your answer.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY