
Concept explainers
(a)
Interpretation:
The major product for the reaction of given
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
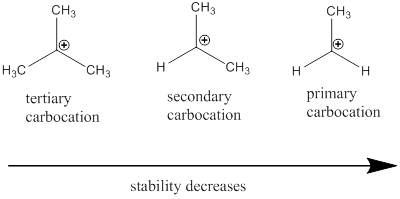
Resonance stabilization: Due to the delocalization of electrons within the molecule the overall energy becomes lower and makes that molecule more stable.
Hydroboration reaction: The reaction involves addition of
(b)
Interpretation:
The major product for the reaction of given alkene under hydro boration should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Resonance stabilization: Due to the delocalization of electrons within the molecule the overall energy becomes lower and makes that molecule more stable.
Hydroboration reaction: The reaction involves addition of
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ORGANIC CHEM ALC PKG
- How is alkene produced by Kolbe’s electrolytic method?arrow_forwardExplain the characteristics of electrophilic addition of HX to alkenes ?arrow_forwarda. Explain why a single pure product is obtained from hydroboration–oxidation of 2-butyne, whereas two products are obtained from hydroboration– oxidation of 2-pentyne. b. Name two other internal alkynes that yield only one product upon hydroboration–oxidation.arrow_forward
- Review the oxidation reactions using Cr6+reagents. Then draw the product formed when compound B is treated with following reagent. CrO3, H2SO4, H2Oarrow_forwardWhich of the following is most soluble in basic medium? a. cyclopropane b. 1,3-cyclobutadiene c. 1,3-cyclopentadiene d. benzenearrow_forwardExplain Halogenation of Activated Benzenes ?arrow_forward
- What changes in color take place when KMnO4 reacts with an alkene?arrow_forwardExplain why the addition of HBr to alkenes A and C is regioselective, forming addition products B and D, respectively.arrow_forwardExplain why the addition of HBr to alkenes A and C is regioselective,forming addition products B and D, respectively.arrow_forward
- make 4-bromo -2nirto aniline from benzene.arrow_forwardDraw the structure of the minor product formed in the reaction of 2-methylbut-1-ene with concentrated sulfuric acid Explain why thid is the major product? Structure of minor product? Explanation?arrow_forwardExplain why a single pure product is obtained from hydroboration–oxidation of 2-butyne, whereas two products are obtained from hydroboration– oxidation of 2-pentyne. a. Name two other internal alkynes that yield only one product upon hydroboration–oxidation.arrow_forward

