
Concept explainers
a)
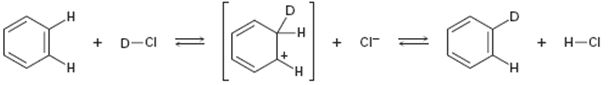
Interpretation:
Curved arrows are to be added to the polar reaction given to indicate the flow of electrons.
Concept introduction:
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To add:
Curved arrows to indicate the flow of electrons in the polar reaction given.
b)
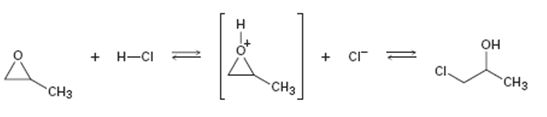
Interpretation:
Curved arrows are to be added to the polar reaction given to indicate the flow of electrons.
Concept introduction:
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To add:
Curved arrows to indicate the flow of electrons in the polar reaction given.
Trending nowThis is a popular solution!

Chapter 6 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- Predict the product(s) of the acid/base reactions below. Draw curved arrows to show the formation and breaking of bonds.arrow_forwardConsider the following reaction: CH4 + Cl• → •CH3 + HCl. Question: Use curved arrows to show the movement of electrons in this radical reaction.arrow_forwardIn the drawing area below, create an acetal with at least 3 methoxy groups, and a total of 5 carbon atoms.arrow_forward
- Please draw the line structures for the C8H17+ carbocations that have the carbon skeleton below And please circle the most stable carbocationarrow_forwardWrite out the mechanism for this hydration of an alkene adding curved arrows to show how electrons reorient to make or break bonds or form new lone pairsarrow_forwardAre terminal alkynes considered weak or strong acids?arrow_forward
- Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Draw the structures of the following compounds. C4H8, an alkane C4H4O2, an esterarrow_forwardPlease help with the following... Draw the bond line structures for the reactants and the products obtained in the following reactions: 1. m-chlorocumene +NaNiPr2, HNiPr2 2. 4-tertbutyl-3-methyl anisole + HNO3, H2SO4 3. o-isopropyl acetophenone + PhCH2COCl, AlCl3arrow_forwardDraw curved arrows to show how the reaction forms the product .arrow_forward
- circle most polar carbon and give partial charges using d- and d+ notationarrow_forwardWhich base is at the 5’ end of the structure shown? A.) A B.) C C.) G D.) T E.) Uarrow_forwardPlease draw the line structures of all of the C8H17+ carbocations that have the carbon-skeleton below. And circle the most stable carbocation of those drawn.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

