
Concept explainers
(a)
Interpretation:
The Lewis electron dot structure for three compounds having molecular formula
Concept Introduction:
- Lewis structures are diagrams that represent the
chemical bonding of covalently bonded molecules and coordination compounds. - It is also known as Lewis dot structures which represent the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
- The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Lewis structure for any molecule is drawn by using the following steps,
First the skeletal structure for the given molecule is drawn then the total number of valence electrons for all atoms present in the molecule is determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed such that each atom contains eight electrons in its valence shell.
(b)
Interpretation:
The geometric isomers have to be identified and labelled.
Concept Introduction:
Cis–trans isomerism (or) geometric isomerism:
In a cis isomer, groups are attached on the same side of the double bond while in the trans isomer the groups are attached on the opposite sides.
Simple representation of cis and Trans isomer is,
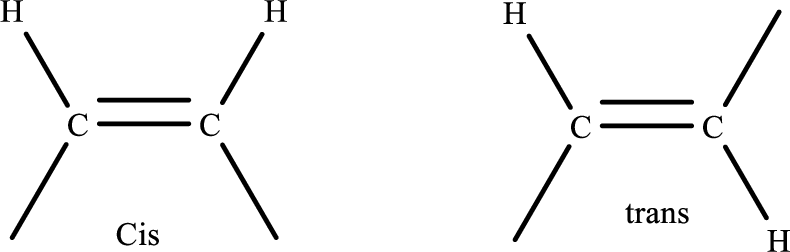 .
.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Chemistry: The Molecular Science
- What structural features help us identify a compound as(a) an alkane, (b) a cycloalkane, (c) an alkene, (d) an alkyne,(e) a saturated hydrocarbon, (f) an aromatic hydrocarbon?arrow_forwardClassify compound as organic or inorganic. C4H6O6arrow_forwardReplace one atom in CH3CH2Br by another Br atom to form a model of a compound with the formula C2H4Br2. Draw projection drawings and perspective drawings of all possible isomers. Does replacing different hydrogen atoms in CH3CH2Br to produce C2H4Br2 give different isomers? (i.e. Are all the hydrogen atoms equivalent?) If there are multiple isomers, what type of isomerism do they represent?arrow_forward
- For the four classes of hydrocarbons (Alkane, Alkene, Alkyne, Arene): Give a structural formula example of each, or identify the characteristic bond that is present.arrow_forwardWhat is the molecular formula of Alkane, Alkene and Alkyne if n=4?arrow_forwardDraw all the isomers of C4H10O using bond-line formulas.arrow_forward
- What is the nature of the bonding in C3H2F2 , Is it polar?arrow_forwardConsider the compound C₂H₃N. Which one of the structures in Figure 4 is the best representation of this compound based on your current knowledge? * A B C D All these structures are good representations of the compound.arrow_forwardStructural isomers have the same molecular formula but have different properties. How is this possible?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning



