
Concept explainers
Ketene, C2H2O, is a reactant for synthesizing cellulose acetate, which is used to make films, fibers, and fashionable clothing.
- (a) Write the Lewis structure of ketene. Ketene does not contain an —OH bond.
- (b) Identify the electron-region geometry and the molecular geometry around each carbon atom and all the bond angles in the molecule.
- (c) Identify the hybridization of each carbon and oxygen atom.
- (d) Is the molecule polar or nonpolar? Use appropriate data to support your answer.
(a)
Interpretation:
Lewis structure of
Concept Introduction:
Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
Explanation of Solution
Given molecule is
The Lewis electron dot structure for given molecule can be determined by first drawing the skeletal structure. Then, the total number of valence electrons for all atoms present in the molecule is determined.
The next step is to subtract the electrons present in the total number of bonds present in the skeletal structure of the molecule with the total valence electrons such that considering each bond contains two electrons with it.
Finally, the electrons which got after subtractions have to be equally distributed considering each atom contains eight electrons in its valence shell.
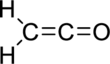
Total number of valence electrons is given below:
Total number of electrons in bonds present is given below:
The four electrons remaining will be distributing in such a way that each atom should have 8 electrons in its outer shell.
Therefore, the Lewis structure is given below:
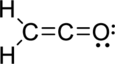
(b)
Interpretation:
The electron-region geometry and the molecular-geometry around each atom and the bond angles in the molecule have to be described.
Concept Introduction:
Electron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone pair of electrons.
Molecular geometry is the shape of a molecule predicted by considering only bond pair of electrons
Geometry of different type of molecules with respect to the number of electron pairs are mentioned below,
Explanation of Solution
Given molecule is
The Lewis electron dot structure is given below:
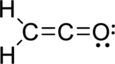 .
.
Consider the first carbon atom.
The molecular geometry and the electron-pair geometry will be trigonal planar because of the presence of three bond pairs around the carbon atom. The
Consider the second carbon atom.
The molecular geometry and the electron-pair geometry will be linear because of the presence of two bond pairs around the carbon atom. The
(c)
Interpretation:
The hybridization of each carbon and oxygen has to be described.
Explanation of Solution
Given molecule is
The Lewis electron dot structure is given below:
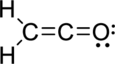 .
.
Consider the first carbon atom.
The molecular geometry and the electron-pair geometry will be trigonal planar because of the presence of three bond pairs around the carbon atom. Therefore, the hybridisation will be
Consider the second carbon atom.
The molecular geometry and the electron-pair geometry will be linear because of the presence of two bond pairs around the carbon atom. Therefore, the hybridisation will be will be
Consider the oxygen atom.
The electron-pair geometry around oxygen will be trigonal planar because of the presence of one bond pair and two lone pair around the oxygen atom. Therefore, the hybridisation will be
(d)
Interpretation:
Whether the molecule is polar or non-polar has to be given.
Explanation of Solution
Given molecule is
The Lewis electron dot structure is given below:
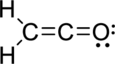 .
.
The
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: The Molecular Science
- Gamma hydroxybutyric acid, GHB, infamous as a date rape drug, is used illicitly because of its effects on the nervous system. The condensed molecular formula for GHB is HO(CH2)3COOH. (a) Write the Lewis structure for GHB. (b) Identify the hybridization of the carbon atom in the CH2 groups and of the terminal carbon. (c) Is hydrogen bonding possible in GHB? If so, write Lewis structures to illustrate the hydrogen bonding. (d) Which carbon atoms are involved in sigma bonds? In pi bonds? (e) Which oxygen atom is involved in sigma bonds? In pi bonds?arrow_forwardA student investigates the physical and chemical properties of various carbon-containing compounds. The complete Lewis electron-dot diagrams and boiling points for two compounds, Q and Z, are shown in the following table: a) Identity the hybridization of the valence orbitals of the carbon atom in compound Q that is indicated by the arrow in the diagram.arrow_forwardDescribe the bonding in formaldehyde (H2CO) in terms of hybrid atomic orbitals and molecular orbitals.arrow_forward
- Draw Lewis structures for pyridine and its conjugate acid, the pyridinium ion, C5H5NH+. What are the geometries and hybridizations about the nitrogen atoms in pyridine and in the pyridinium ion?arrow_forwardFor a N H 4 + ion, identify its molecular shape, bond angle, and hybrid orbitals.arrow_forwardA 3D representation of a cyclohexane (C6 H 12) molecule, a cyclic compound used in the manufacture of nylon and found in the distillation of petroleum, is shown. Name the geometry around each carbon atom. geometry: What is the hybridization of each carbon atom?arrow_forward
- Explain the structure of benzene (C6H6) in detail using a ubiquitous electron model (the theory of atomic electron bonding) and the theory of molecular orbital function theoryarrow_forwardDraw the Lewis structure for 1, 1-dimethylhydrazine [(CH3)2NNH2, a compound used as a rocket fuel]. What: is the hybridization for the two nitrogen atoms in this molecule? What orbitals overlap to form the bond between the nitrogen atoms?arrow_forwardConsider the polyatomic ion IO65-. How many pairs of electrons are around the central iodine atom? What is its hybridization? Describe the geometry of the ion.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




