
Concept explainers
Figure 7.11 Dinitrophenol (DNP) is an "uncoupler" that makes the inner mitochondrial membrane "leaky" to protons. It was used until 1938 as a weight-loss drug. What effect would you expect DNP to have on the change in pH across the inner mitochondrial membrane? Why do you think this might be an effective weight-loss drug?
To write:
The effect of DNP on the pH across the mitochondrial membrane and the reason that it is a weight-loss drug.
Introduction:
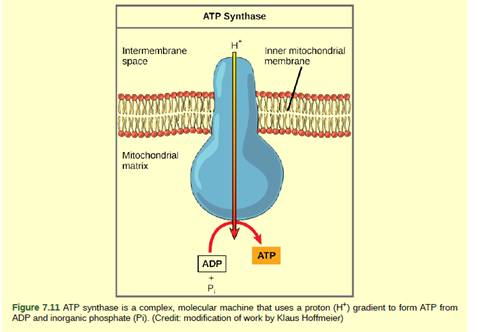
Dinitrophenol (DNP) is an uncoupler. The flow of electrons can be separated by DNP along with the pumping of the H+ ion for the purpose of production of ATP (adenosine triphosphate). It means that the electron transport chain can no longer form a proton gradient, and ATP synthase can no longer make ATP.
DNP is a drug given to the patient for losing weight. After using DNA as weight loss drug, a person obtains less energy out of the eaten food. One of the worst side effects of taking DNP is overheating of the body, the energy from electron transport is lost as heat.
Explanation of Solution
DNP is an uncoupler, it disrupts the ATP synthesis by leaking protons across the inner mitochondrial membrane. As a result, the proton gradient cannot be formed across the inner mitochondrial membrane as pumping of H+ (hydrogen ion) is stopped. So, there will be a decrease in pH across the inner membrane of mitochondria.
Therefore, ATP production becoming less efficient. Hence, the energy which is normally produced during cellular respiration is being wasted as heat. In such condition, majority of the eaten food could not be used for the purpose of ATP synthesis and we lose weight.
DNP is an effective diet drug used to lose weight. It acts as an uncoupler which disrupts the H+ gradient across mitochondrial membrane reducing ATP synthesis. Since ATP cannot be formed, the energy from electron transport is lost as heat.
Want to see more full solutions like this?
Chapter 7 Solutions
EBK BIOLOGY
Additional Science Textbook Solutions
College Physics
Campbell Biology in Focus
Concepts of Genetics (11th Edition)
Concepts of Genetics (12th Edition)
Human Physiology: An Integrated Approach (7th Edition)
Campbell Essential Biology with Physiology (6th Edition)
- Figure 4.15 Cyanide inhibits cytochrome c oxidase, a component of the electron transport chain. If cyanide poisoning occurs, would you expect the pH of the intermembrane space to increase or decrease? What affect would cyanide have on ATP synthesis? Figure 4.15 (a) The electron transport chain is a set of molecules that supports a series of oxidation-reduction reactions. (b) ATP synthase is a complex, molecular machine that uses an H+ gradient to regenerate ATP from ADP. (c) Chemiosmosis relies on the potential energy provided by the H+ gradient across the membrane.arrow_forwardWhich of the following statements is a lie? Select one: a.The formation of water aids in establishing the ion gradient between the inner matrix and intermembrane space. b.Every protein complex in the electron transport chain is involved in the direct pumping of H+ in the intermembrane space. c.There is an existing electrochemical gradient between the intermembrane space and the inner mitochondrial matrix.arrow_forwardWhy is the enzyme-catalyzed introduction of carbon–carbon double bonds into fatty acids called an electron transport system?arrow_forward
- What would be the effect on ATP production of decreasing the pH inside the mitochondrial matrix? Explainarrow_forwardWhich electrically-neutral molecules are generated when complex IV (of the mitochondrial electron transport chain) adds 4 electrons and 4 protons to one molecule of diatomic oxygen gas? two molecules of the superoxide free radical two molecules of hydrogen peroxide two molecules of water two molecules of the hydroxide ion two molecules of the hydroxyl free radicalarrow_forwardWhich of the following components of the mitochondrial electron transport chain is the only one to be considered a peripheral protein? the cytochrome a-a3 complex the cytochrome c oxidase complex coenzyme Q (ubiquinone) the cytochrome b-c1 complex cytochrome carrow_forward
- Which of the following components of the mitochondrial electron transport chain is the only one to be considered a peripheral protein? cell biology the cytochrome a-a3 complex the cytochrome c oxidase complex coenzyme Q (ubiquinone) the cytochrome b-c1 complex cytochrome carrow_forwardCHOOSE THE CORRECT LETTER Which of the following conditions promote the synthesis of ATP through the electron transport chain?A.proton pump from outer mitochondrial membrane to inner mitochondrial membraneB. proton pump from matrix to inner mitochondrial membraneC. proton pump from inner mitochondrial membrane to outer mitochondrial membraneD. proton pump from inner mitochondrial membrane to matrixarrow_forwardWhich of the following electron carriers diffuses within the inner mitochondrial membrane to transport electrons? A. Cytochrome c B. Flavoproteins C. Fe-S Cluster Proteins D. Ubiquinolarrow_forward
- What are two advantages of the components of the electron transport chain being embedded in the inner mitochondrial membrane?arrow_forwardWhich of the following components of the mitochondrial electron transport chain is not integral to the inner mitochondrial membrane (of a typical eukaryotic cell)? the cytochrome c oxidase complex coenzyme Q (ubiquinone) the cytochrome b-c1 complex cytochrome c the cytochrome a-a3 complexarrow_forwardDescribe the involvements of the followings in the electron transport centre in the inner mitochondrial membrane.(a). Rieske center(b). Ubiquinone(c). Protonsarrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning


