
Interpretation:
The compounds having delocalized electrons have to be predicted.
Concept Introduction:
Resonance Contributor: The appropriate structure with the delocalized electrons is called a resonance contributor, a resonance structure, or a contributing resonance structure.
Delocalized electrons: The sharing of electrons between two or more atoms known as delocalization of electrons. In order to have delocalized electrons, the system must be planar and have alternative double bonds and single bonds.
Resonance hybrid: The actual structure with delocalized electrons is called a resonance hybrid.
Explanation of Solution
Reason for correct options:
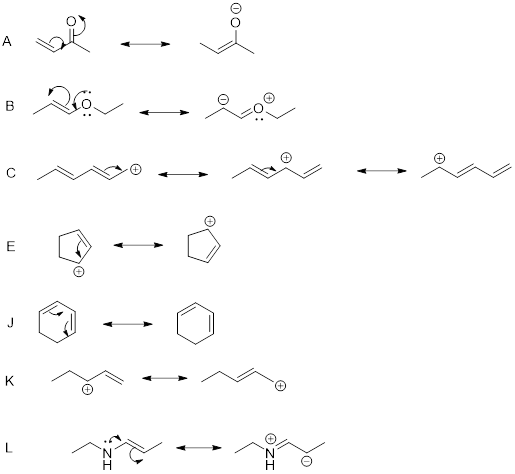
The above compounds all have delocalized electrons, which undergo electron movement.
The resonance structures can be drawn as shown above.
Hence, the correct options are A, B, C, E, J, K and L.
Reason for in-correct options:
The remaining compounds do not contain delocalized electrons since the conjugated system is absent.
Hence, the in-correct options are D, F, G, H, I, J, and K.
The compounds containing delocalized electrons were predicted.
Want to see more full solutions like this?
Chapter 7 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- 1. Identify the relationship between the following two structures.a. Identicalb. neitherc. diastereomers2. Identify the relationship between the following two structures.a. enantiomersb. diastereomersarrow_forwardCH3CH₂CH₂CCI AICI3 A HNO3 H₂SO4 Barrow_forwardWhich compounds are meso compounds?arrow_forward
- Which molecule is optically active? Click on a letter A through D to answer. CH3 Br CI NH2 Br A. В. C. D.arrow_forwardAssign an R or S configuration to the chiral center in each molecule.arrow_forwardWhat is the relationship between these two compounds ? xx a. same compounds b, constitutional isomers OC. Enantiomers Od. Diastereomersarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
