
Concept explainers
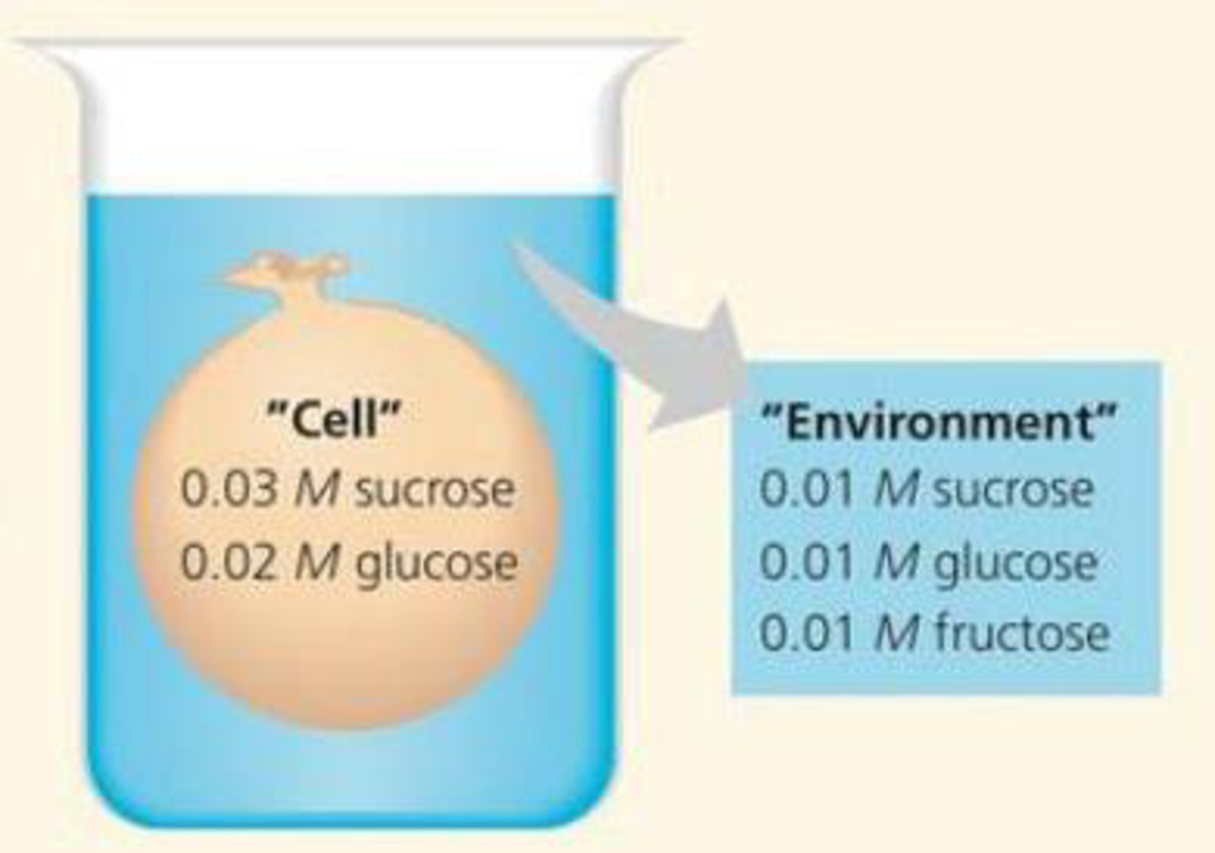
DRAW IT An artificial "cell" consisting of an aqueous solution enclosed in a selectively permeable membrane is immersed in a beaker containing a different solution, the “environment”, as shown in the accompanying diagram. The membrane is permeable to water and to the simple sugars glucose and fructose but impermeable to the disaccharide sucrose.
- (a) Draw solid arrows to indicate the net movement of solutes into and/or out of the cell.
- (b) Is the solution outside the cell isotonic, hypotonic, or hypertonic?
- (c) Draw a dashed arrow to show the net osmosis, if any.
- (d) Will the artificial cell become more flaccid, more turgid, or stay the same?
- (e) Eventually, will the two solutions have the same or different solute concentrations?
Learn your wayIncludes step-by-step video

Chapter 7 Solutions
Campbell Biology, Books a la Carte Plus Mastering Biology with Pearson eText -- Access Card Package (11th Edition)
Additional Science Textbook Solutions
Campbell Essential Biology (7th Edition)
Brock Biology of Microorganisms (14th Edition)
Campbell Biology: Concepts & Connections (8th Edition)
Microbiology: An Introduction (13th Edition)
Biological Science (6th Edition)
Microbiology with Diseases by Body System (4th Edition)
- 41. A TRANSPORT MECHANISM ACROSS A MEMBRANE WHERE THE MOVEMENT OF THE SOLUTION IS FROM HIGHER TO LOWER CONCENTRATION UNTIL SUCH TIME THAT EQUILIBRIUM IS ESTABLISHED?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 42. BIOMOLECULES ARE HYDROLYZED FIRST INTO THEIR SIMPLEST FORM BEFORE THEY CAN BE ABSORBED BY THE TISSUES. GLUCOSE ABSORPTION IS THROUGH WHICH OF THE FOLLOWING?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 43. A TRANSPORT MECHANISM THROUGH MEMBRANE WHICH NEEDS ATP?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 44. TRANSPORT MECHANISM ACROSS MEMBRANE OF GASES?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSIS 45. A TRANSPORT MECHANISM IN WHICH SMALLER PARTICLES PASSES THROUGH MEMBRANE BUT NOT THE BIG ONE?A. OSMOSISB. FREE DIFFUSIONC. FACILITATED ABSORPTIOND. ACTIVE TRANSPORTE. DIALYSISarrow_forwardCreate a diagram that demonstrates the difference between tonicity and osmolarity. Be sure to label the new solutions in each condition, draw the changes (if predicted) in cell shape/volume, and use arrows to show the movement of water relative to the cell. Please start with a red blood cell in an isosmotic solution and show what happens to cell shape and water movement when NaCl is added to the solution, or when water is added.arrow_forwardAssume a cell has an osmolarity of 10 mM. What will happen to that cell (swell, shrink, or stay the same) if it is placed in a solution of: a) 5 mM glucose b) 10 mM glucose c) 10 mM NaClarrow_forward
- Osmosis is often defined as the flow of water through a semipermeable membrane, from a dilute solution to a more concentrated solution. Video experiment #1 – saturated sucrose/dialysis tubing/water osmosis from U Mich. https://youtu.be/pCupvFGN4bw A saturated solution of sucrose-containing a red dye is separated from water by dialysis material. What happens to the volume of sucrose solution during the experiment? How do you know that the sucrose solution did not flow into the beaker of water? (Give evidence from your observation of the experiment.) Diffusion, Osmosis, Dialysis video: https://youtu.be/tHzkRtzVmUMDialysis, the flow of both solvent and small molecules or ions through a dialyzing membrane, is important clinically in the operation of the artificial kidney machine.arrow_forwardhosphate buffered saline (PBS) is a buffer solution commonly used in biological The buffer helps to maintain a constant pH. The osmolality and ion concentrations of the solution usually match those of the human body. You need to prepare a stock solution at pH 7.00 with NaH2PO4 and Na2HPO4 (pKa =7.21). What would be the respective concentration of these substances if you wish to obtain the final phosphate concentration [HPO4 −2] + [H2PO4 −] = 0.3 M? What will be the pH of the solution after adding 0.18 L of 0.2 M NaOH? What is the ratio of HPO4−2 of H2PO4 – is part b, and what is the significance? NON GRADED PRACTICE QUESTION FOR BIOCHEMarrow_forwardGiven the following cell type and solutions, draw the cell, showing the net direction of osmosis (water flow) and say what state the cell will be in. Choose from the following terms to describe the state of the cell: Turgid, flaccid, plasmolyzed, crenulated (shriveled), lysed (burst), normal. An animal cell with an internal osmolarity of 0.32 M (total concentration of all the solutes in solution) that is placed in a solution of 0.32 M NaCl.arrow_forward
- Predict the fate of a red blood cell placed in each of the solutions below. Explain why these changes would take place. (Calculate osmolarity for each solution). Take into account that some will ionize and some will not. 0.3 M Glucose 0.3 M NaCl 0.15 M NaClarrow_forwardA phospholipid bilayer separates two chambers. The chambers both contain the same volume of water. The left chamber contains 150 mM of an unknown solute A. Solute B is dissolved in the right chamber also at a concentration of 150 mM. Therefore, the initial osmolarity is the same (isotonic) between the two chambers. However, after a short time, the volume increases substantially in the right chamber. What is the most likely ranking of permeability for the molecules to cause this change? (greater permeability means that the molecule moves first across the membrane)arrow_forward“Plasma membrane is described as protein iceberg in sea of lipids”. why ?arrow_forward
- Explain how the following affect membrane fluidity:– Level of phospholipid tail saturation– Level of cholesterol– Phospholipid tail lengtharrow_forwardAn artificial cell consisting of an aqueous solution enclosed in a selectively permeable membrane (but with no cell wall) is immersed in a beaker containing an aqueous solution. The outside environment concentration consists of 0.01 M glucose and the inside of the cell has a concentration of 5.0 M glucose. The plasma membrane is permeable to water and monosaccharides, but impermeable to the disaccharides. Complete the following for the image below: Is the glucose going down or against its concentration gradient? Is the movement of the solute in the cell going out of the cell? Down...Yes Down...No Against...Yes Against...Noarrow_forwardPhospholipids are synthesized within the cytosolic half of the smooth ER membrane bilayer. true falsearrow_forward
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning

